Jazz Pharmaceuticals Announces Second Quarter 2021 Financial Results
- Closed GW Pharmaceuticals plc Acquisition, Creating an Innovative, High-Growth, Global Biopharma Leader
- 41% of Net Product Sales from Recently Launched or Acquired Products
- Strong Xywav™ Adoption with 5,100 Active Patients Exiting the Second Quarter
- FDA Approval and Launch of Rylaze™ in the U.S.
- Expect to Initiate Pivotal Phase 3 Trial of Epidiolex® for Epilepsy with Myoclonic-Atonic Seizures in First Half 2022
- Total Revenues Increased 34% to $751.8 Million Compared to Second Quarter 2020
- 2021 Total Revenue Guidance Affirmed at $3.02 Billion to $3.18 Billion
DUBLIN, Aug. 3, 2021 /PRNewswire/ — Jazz Pharmaceuticals plc (Nasdaq: JAZZ) today announced financial results for the second quarter of 2021 and affirmed non-GAAP adjusted financial guidance for 2021.
As we enter what we expect to be a period of sustained growth, I have never been more excited about the future for Jazz. The recent approval and launches of Xywav and Rylaze exemplify Jazz today.
 Bruce Cozadd, chairman and chief executive officer of Jazz Pharmaceuticals
Bruce Cozadd, chairman and chief executive officer of Jazz Pharmaceuticals
We are rapidly establishing ourselves as an innovative biopharmaceutical company with expanding R&D capabilities and substantial commercial prowess, underscored by our consistent execution across the business. The addition of the GW cannabinoid platform and related pipeline complement and enhance our own growing R&D capabilities, accelerating our ability to improve the lives of patients.
Cozadd added, “We have now executed four of five planned product launches since the beginning of 2020 and look forward to our anticipated launch of Xywav in idiopathic hypersomnia later this year, a critical step forward for these underserved patients. With 41% of our second quarter net product sales from recently launched or acquired products, we are well on track to meet our revenue diversification targets while driving significant shareholder value.”
Robert Iannone, M.D., M.S.C.E., executive vice president, research and development and chief medical officer, added, “We are excited that Rylaze was recently approved in the United States and is now broadly available to acute lymphoblastic leukemia and lymphoblastic lymphoma patients in critical need. We aim to further leverage our proven R&D capabilities to deliver on the significant value of our pipeline and the GW cannabinoid platform. The shared values and patient-centricity among the Jazz and GW teams, coupled with the successful ongoing integration, will further enhance our ability to innovate and execute, including the planned initiations of a Phase 3 pivotal trial for Epidiolex in epilepsy with myoclonic-atonic seizures and the third Phase 3 nabiximols clinical trial in multiple sclerosis-related spasticity.”
Business Updates
Corporate Development
On May 5, 2021, the Company completed the acquisition of GW Pharmaceuticals plc (GW) for a total value of approximately $7.2 billion, or $6.8 billion net of GW cash. The Company secured $5.35 billion of financing to fund the GW transaction. The financing structure supports the Company’s plans for rapid deleveraging to its stated targets while also continuing to make investments to grow the business. The combined company is a leader in neuroscience with a global commercial and operational footprint, well positioned to maximize the value of its diversified portfolio.
Neuroscience
Oxybate (Xyrem® and Xywav):
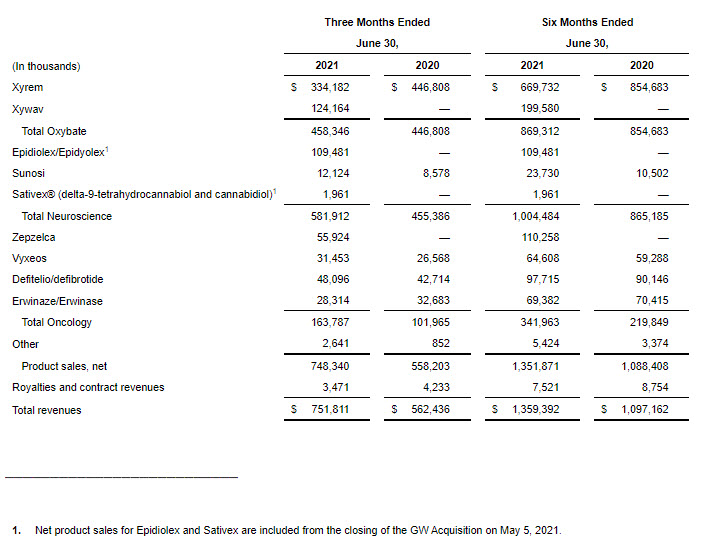
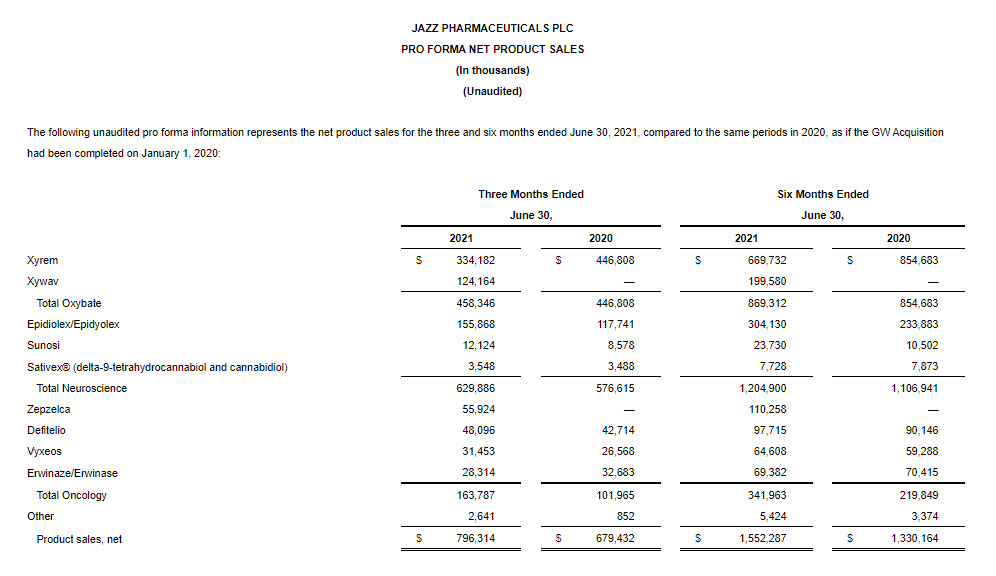
- Net product sales for the combined oxybate business increased 3% to $458.3 million in the second quarter of 2021 compared to the same period in 2020.
- Average active oxybate patients on therapy were approximately 15,900 in the second quarter of 2021, an increase of approximately 5% compared to the same period in 2020.
Xywav (calcium, magnesium, potassium, and sodium oxybates) oral solution:
- Xywav net product sales were $124.2 million in the second quarter of 2021.
- There were approximately 5,100 active patients on Xywav exiting the second quarter of 2021.
- In June 2021, FDA recognized seven years of Orphan Drug Exclusivity for Xywav.
- FDA published its summary of clinical superiority findings for Xywav stating that Xywav is clinically superior to Xyrem by means of greater safety because Xywav provides a greatly reduced chronic sodium burden compared to Xyrem, and that the differences in the sodium content of the two products at the recommended doses will be clinically meaningful in reducing cardiovascular morbidity in a substantial proportion of patients for whom the drug is indicated.
- The Company has achieved its goal of obtaining broad payer coverage, having entered into agreements with all three of the largest pharmacy benefit managers.
Xyrem (sodium oxybate) oral solution:
- Xyrem net product sales decreased 25% to $334.2 million in the second quarter of 2021 compared to the same period in 2020.
Xywav in Idiopathic Hypersomnia
- FDA has granted Priority Review Designation and accepted the supplemental New Drug Application (sNDA) for Xywav in adult patients with idiopathic hypersomnia (IH). The Prescription Drug Fee User Act (PDUFA) target date for an FDA decision has been set for August 12, 2021, which is in line with the Company’s objective of launching in the fourth quarter of 2021 following risk evaluation and mitigation strategy (REMS) implementation.
Epidiolex/Epidyolex® (cannabidiol):
- Epidiolex/Epidyolex net product sales were $109.5 million in the second quarter of 2021. This includes sales from May 5, 2021, the closing date of the GW Acquisition.
- On an unaudited pro forma basis, net product sales in the second quarter of 2021 increased by 32% to $155.9 million compared to the same period in 2020.
- The Company expects to initiate a Phase 3 pivotal trial of Epidiolex for Epilepsy with Myoclonic-Atonic Seizures (EMAS), also known as Doose syndrome, in the first half of 2022. EMAS represents the fourth target indication for Epidiolex.
Sunosi® (solriamfetol):
- Sunosi net product sales increased by 41% to $12.1 million in the second quarter of 2021 compared to the same period of 2020.
- In the second quarter of 2021, U.S. prescriptions increased by 25% compared to the first quarter of 2021.
Nabiximols:
- The Company expects to initiate the third Phase 3 nabiximols clinical trial in multiple sclerosis (MS)-related spasticity this year.
- The two ongoing Phase 3 clinical trials in MS-related spasticity continue to progress.
JZP385:
- JZP385, a highly selective modulator of T-type calcium channels, is in clinical development for the potential treatment of essential tremor.
- The Company expects to initiate a Phase 2b trial in late 2021.
JZP150:
- JZP150, a fatty acid amide hydrolase (FAAH) inhibitor, is in clinical development for the potential treatment of post-traumatic stress disorder.
- The Company expects to initiate a Phase 2 trial in late 2021.
Oncology
Zepzelca™ (lurbinectedin):
- Zepzelca net product sales were $55.9 million in the second quarter of 2021.
- Sequential demand growth over the first two quarters of 2021 was 8% and 9% respectively, offset mainly by reduced inventory holding by distributors.
- Robust Zepzelca development program planned:
- The Company’s partner, PharmaMar, plans to initiate a confirmatory trial in second-line small cell lung cancer (SCLC) later this year. If positive, this trial would confirm the benefit of Zepzelca in the treatment of SCLC when patients progress following first-line treatment with a platinum-based regimen.
- The Company is collaborating with Roche to initiate a Phase 3 pivotal clinical trial in first-line extensive stage SCLC in combination with immunotherapy this year.
- The Company expects to initiate a Phase 2 basket trial in early 2022 to explore lurbinectedin monotherapy in patients with select advanced or metastatic solid tumors. Cohorts will include advanced urothelial cancer, large cell neuroendocrine tumor of the lung, and homologous recombinant deficient positive (HRD+) cancers.
- The Company has initiated a Phase 4 observational study to collect real world safety and outcome data in adult Zepzelca monotherapy patients with extensive stage small cell lung cancer who progress on or after prior platinum-containing chemotherapy.
Rylaze (asparaginase erwinia chrysanthemi (recombinant)-rywn):
- On June 30, 2021, FDA approved Rylaze under the Real-Time Oncology Review program for use as a component of a multi-agent chemotherapeutic regimen for the treatment of acute lymphoblastic leukemia (ALL) or lymphoblastic lymphoma (LBL) in pediatric patients one month and older and adult patients who have developed hypersensitivity to E. coli-derived asparaginase.
- Rylaze was launched and commercially available in the U.S. on July 15, 2021.
- Rylaze is the only recombinant Erwinia asparaginase manufactured product that maintains a clinically meaningful level of asparaginase activity throughout the entire duration of treatment. It was developed by Jazz to address the needs of patients and healthcare providers for an innovative, high-quality Erwinia asparaginase with reliable supply.
- Rylaze was granted orphan drug designation for the treatment of ALL/LBL by FDA in June 2021.
- The Company will continue to work with FDA and plans to submit additional data in support of a Monday/Wednesday/Friday dosing schedule. Part B of the study is evaluating intravenous administration and is ongoing. The company also plans to submit this data for presentation at a future medical meeting.
- The Company anticipates that data from the current development program will support regulatory filings in Europe in 2022 and is currently working with an in-country partner to advance the program for filing, approval and launch in Japan.
Vyxeos® (daunorubicin and cytarabine) liposome for injection:
- Vyxeos net product sales increased 18% to $31.5 million in the second quarter of 2021 compared to the same period in 2020.
Defitelio® (defibrotide sodium) / defibrotide:
- Defitelio/defibrotide net product sales increased 13% to $48.1 million in the second quarter of 2021 compared to the same period in 2020.
Erwinaze® / Erwinase® (asparaginase Erwinia chrysanthemi):
- Erwinaze/Erwinase net product sales decreased 13% to $28.3 million in the second quarter of 2021 compared to the same period in 2020.
- The Company’s agreement with Porton Biopharma Limited terminated on December 31, 2020. The Company had the right to sell certain Erwinaze inventory post-termination. Sale of this inventory was completed in June 2021.
Financial Highlights
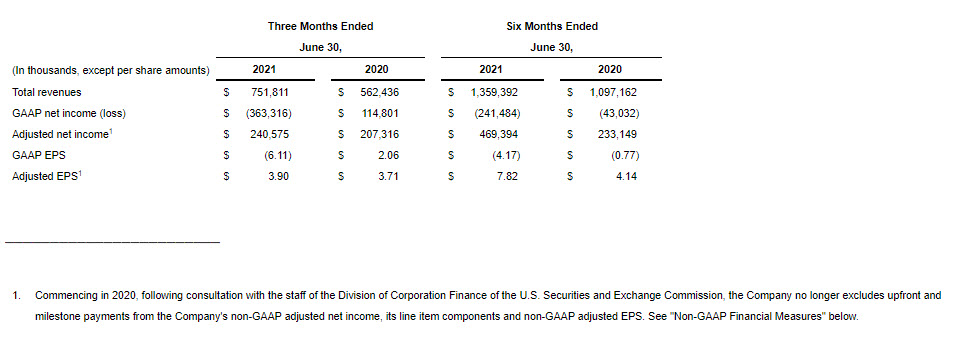
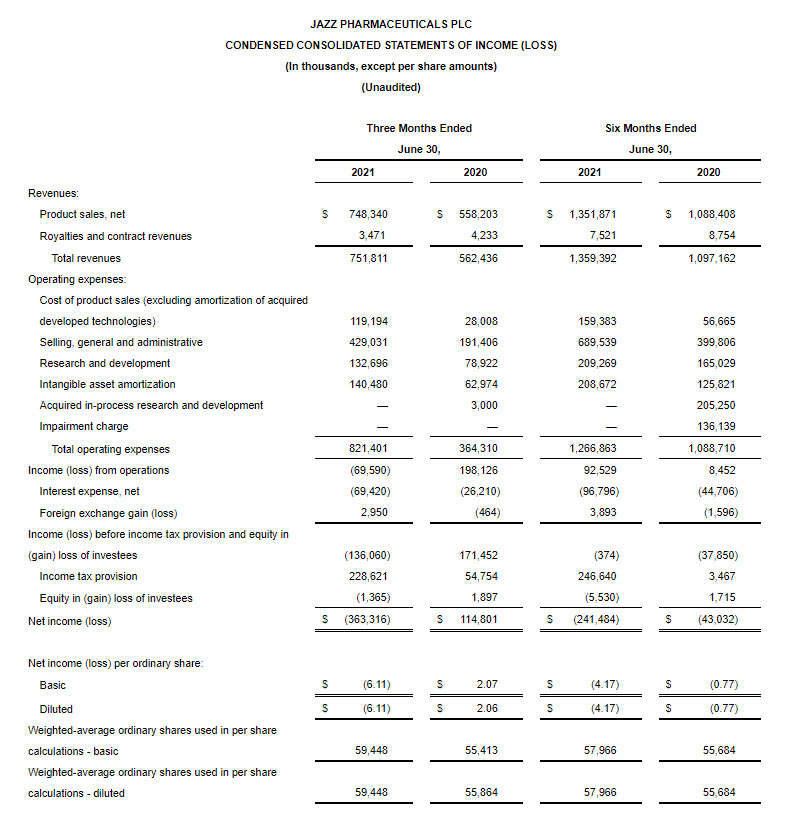
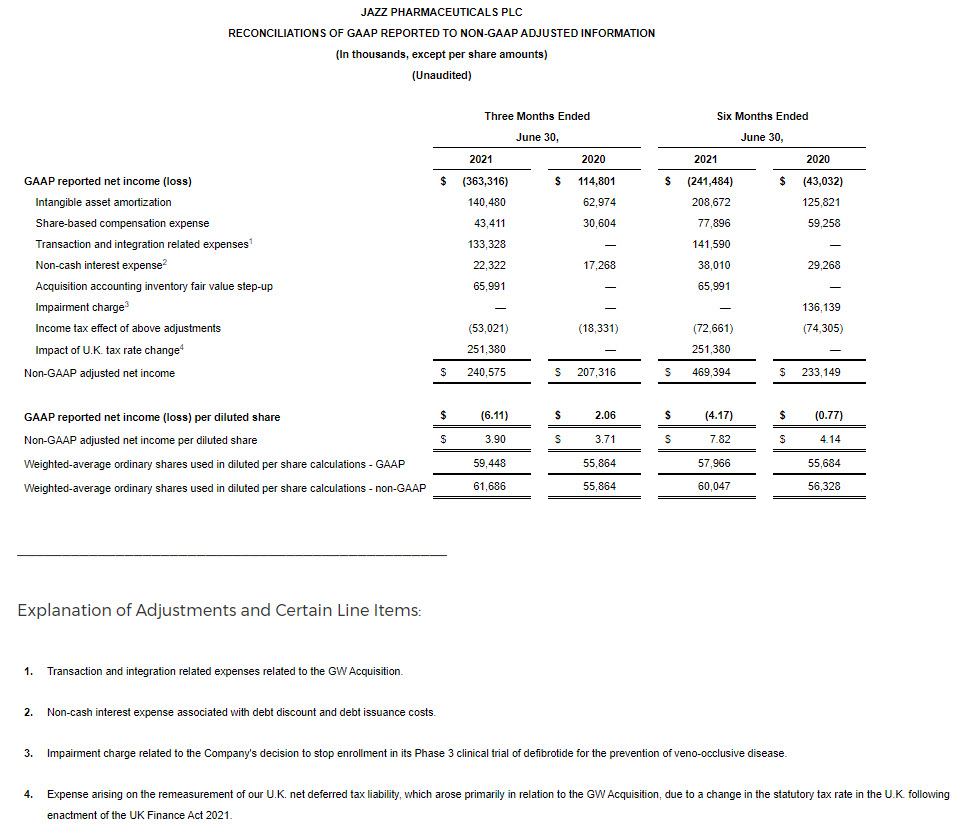
GAAP net income (loss) for the second quarter of 2021 was ($363.3 million), or ($6.11) per diluted share, compared to $114.8 million, or $2.06 per diluted share, for the second quarter of 2020.
Non-GAAP adjusted net income for the second quarter of 2021 was $240.6 million, or $3.90 per diluted share, compared to $207.3 million, or $3.71 per diluted share, for the second quarter of 2020.
Reconciliations of applicable GAAP reported to non-GAAP adjusted information are included at the end of this press release.
Total Revenues
Total revenues increased 34% in the second quarter of 2021 compared to the same period in 2020.
- Products launched or acquired since 2019 accounted for 41% of total net product sales in the second quarter of 2021.
- Neuroscience net product sales in the second quarter of 2021 increased 28% to $581.9 million compared to the same period in 2020. Oxybate net product sales increased to $458.3 million led by strong Xywav net product sales of $124.2 million partially offset by a decrease in Xyrem net product sales as a result of the strong adoption of Xywav by existing Xyrem patients. Epidiolex/Epidyolex net product sales from the date of acquisition were $109.5 million.
- Oncology net product sales in the second quarter of 2021 increased 61% to $163.8 million compared to the same period in 2020 primarily driven by robust Zepzelca net product sales of $55.9 million. Zepzelca launched in the U.S. in July 2020.
Operating Expenses and Effective Tax Rate
Operating expenses changed over the prior year periods primarily due to the following:
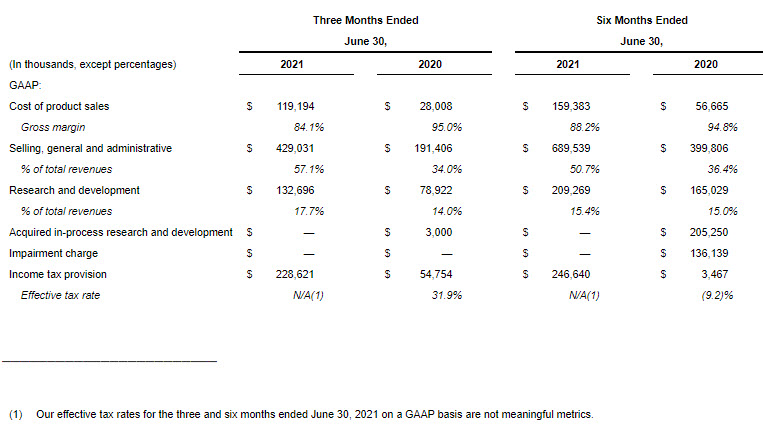
- Cost of product sales increased in the second quarter of 2021 compared to the same period in 2020, on a GAAP and non-GAAP adjusted basis, primarily due to increased net product sales as a result of the GW Acquisition. In addition, an acquisition accounting inventory fair value step-up expense of $66.0 million impacted GAAP cost of product sales.
- Selling, general and administrative (SG&A) expenses increased in the second quarter of 2021 compared to the same period in 2020, on a GAAP and on a non-GAAP adjusted basis, primarily due to an increase in compensation-related expenses driven by higher headcount as a result of the GW Acquisition, increased investment in sales, marketing and launch activities primarily related to Sunosi, Xywav and Zepzelca in the U.S. and the addition of costs related to Epidiolex. SG&A expenses in the second quarter of 2021 on a GAAP basis also included transaction and integration related expenses of $129.5 million related to the GW Acquisition.
- Research and development expenses increased in the second quarter of 2021 compared to the same period in 2020, on a GAAP and on a non-GAAP adjusted basis, primarily due to the addition of costs related to clinical programs for Epidiolex, nabiximols and cannabinoids, an increase in costs for JZP385 and an increase in compensation-related expenses due to higher headcount primarily driven by the GW Acquisition.
- On a GAAP basis, our income tax provision for the three months ended June 30, 2021, included an expense of $251.4 million arising on the remeasurement of our U.K. net deferred tax liability, which arose primarily in relation to the GW Acquisition, due to a change in the statutory tax rate in the U.K. following enactment of the UK Finance Act 2021. Due to the impact of this expense, our effective tax rate for the three months ended June 30, 2021, on a GAAP basis is not a meaningful metric.
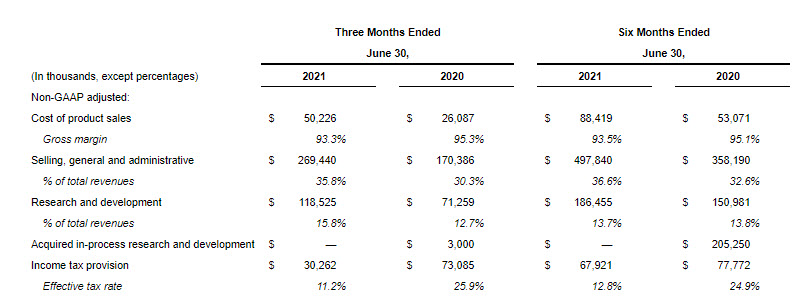
- On a non-GAAP basis, the decrease in the effective tax rate in the second quarter of 2021 compared to the same period in 2020 was primarily due to the impact in 2020 of the disallowance of certain interest deductions, provision for the settlement reached with the French tax authorities, and the impact of the change in income mix.
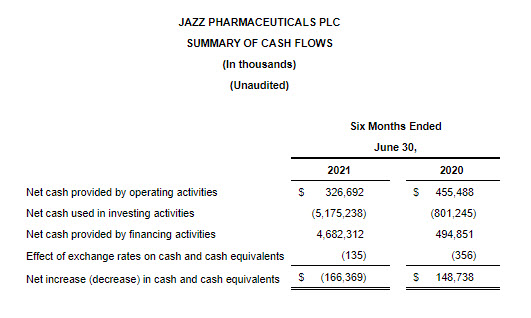
Cash Flow and Balance Sheet
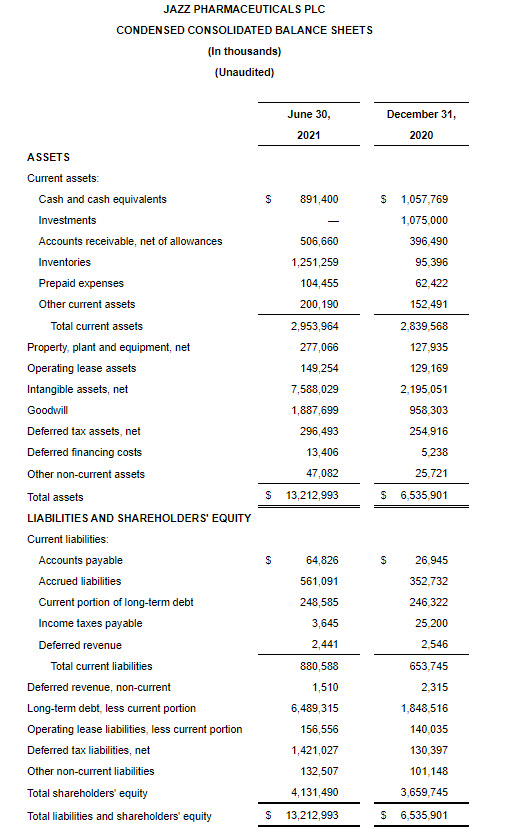
As of June 30, 2021, cash and cash equivalents were $891.4 million, and the outstanding principal balance of the Company’s long-term debt was $7.1 billion. In addition, the Company had undrawn borrowing capacity under a revolving credit facility of $500.0 million.
For the six months ended June 30, 2021, the Company generated $326.7 million of cash from operations.
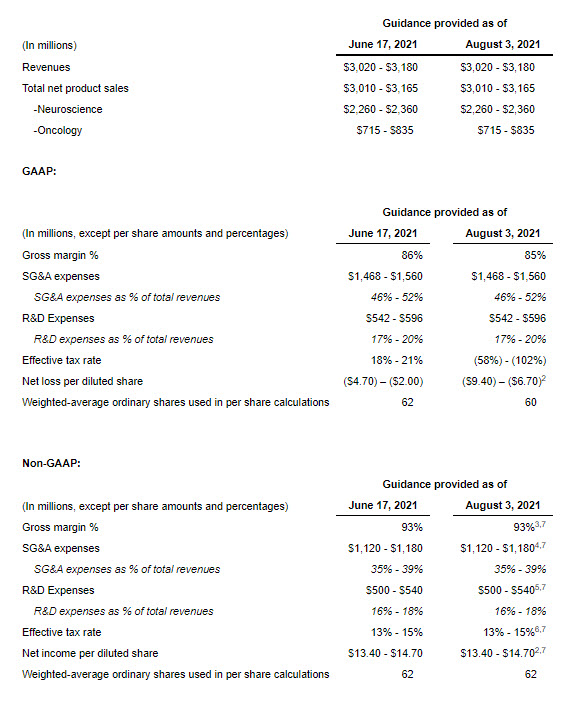
2021 Financial Guidance¹
Jazz Pharmaceuticals is reaffirming its previously communicated full year 2021 non-GAAP financial guidance and updating its 2021 GAAP guidance. This guidance reflects the Company’s current and future expected operational performance, including COVID-19 related impacts, the strength of its underlying operations and the prioritization of new and ongoing value creating development projects.
____________________________
- The Company’s 2021 financial guidance includes the anticipated results of the acquired GW business from the date of acquisition (May 5, 2021) and related acquisition accounting adjustments, which are subject to change if the company obtains additional information during the measurement period (up to one year from the acquisition date); any such change could be material.
- The Company expects the transaction to be dilutive to both GAAP and non-GAAP adjusted net income per diluted share in 2021. On a GAAP basis, this is expected to be primarily due to an increase in the amortization of acquisition-related intangible assets and transaction and integration related expenses, the amortization of inventory fair value step-up, increased interest expense and an increase in number of outstanding shares relating to the GW Acquisition. On a non-GAAP adjusted basis, this is expected to be due to increased cash interest expense and an increase in the number of outstanding shares.
- Excludes $220-$260 million of amortization of acquisition-related inventory fair value step-up, $8-$10 million of share-based compensation expense and $2-$4 million of transaction and integration related expenses relating to the GW Acquisition from estimated GAAP gross margin.
- Excludes $221-$245 million of transaction and integration related expenses relating to the GW Acquisition and $127-$135 million of share-based compensation expense from estimated GAAP SG&A expenses.
- Excludes $35-$45 million of share-based compensation expense and $7-$11 million of transaction and integration related expenses relating to the GW Acquisition from estimated GAAP R&D expenses.
- Excludes the income tax effect of adjustments between GAAP net income and non-GAAP adjusted net income and an expense of $251 million arising on the remeasurement of our U.K. net deferred tax liability, which arose primarily in relation to the GW Acquisition, due to a change in the statutory tax rate in the U.K. following enactment of the UK Finance Act 2021.
- See “Non-GAAP Financial Measures” below. Reconciliations of non-GAAP adjusted guidance measures are included above and in the table titled “Reconciliation of GAAP to Non-GAAP Adjusted 2021 Net Income Guidance” at the end of this press release.
Conference Call Details
Jazz Pharmaceuticals will host an investor conference call and live audio webcast today at 4:30 p.m. ET (9:30 p.m. IST) to provide a business and financial update and discuss its 2021 second quarter results. The live webcast may be accessed from the Investors section of the Company’s website at www.jazzpharmaceuticals.com. Please connect to the website prior to the start of the conference call to ensure adequate time for any software downloads that may be necessary. Investors may participate in the conference call by dialing +1 855 353 7924 in the U.S., or +1 503 343 6056 outside the U.S., and entering passcode 7187077.
A replay of the conference call will be available through August 10, 2021 by dialing +1 855 859 2056 in the U.S., or +1 404 537 3406 outside the U.S., and entering passcode 7187077. An archived version of the webcast will be available for at least one week in the Investors section of the Company’s website at www.jazzpharmaceuticals.com.
About Jazz Pharmaceuticals
Jazz Pharmaceuticals plc (NASDAQ: JAZZ) is a global biopharmaceutical company whose purpose is to innovate to transform the lives of patients and their families. We are dedicated to developing life-changing medicines for people with serious diseases – often with limited or no therapeutic options. We have a diverse portfolio of marketed medicines and novel product candidates, from early- to late-stage development, in neuroscience and oncology. Within these therapeutic areas, we are identifying new options for patients by actively exploring small molecules and biologics, and through innovative delivery technologies and cannabinoid science. Jazz is headquartered in Dublin, Ireland and has employees around the globe, serving patients in nearly 75 countries. For more information, please visit www.jazzpharmaceuticals.com and follow @JazzPharma on Twitter.
Non-GAAP Financial Measures
To supplement Jazz Pharmaceuticals’ financial results and guidance presented in accordance with U.S. generally accepted accounting principles (GAAP), the Company uses certain non-GAAP (also referred to as adjusted or non-GAAP adjusted) financial measures in this press release and the accompanying tables. In particular, the Company presents non-GAAP adjusted net income (and the related per share measure) and its line item components, as well as certain non-GAAP adjusted financial measures derived therefrom, including non-GAAP adjusted gross margin percentage and non-GAAP adjusted effective tax rate. Non-GAAP adjusted net income (and the related per share measure) and its line item components exclude from GAAP reported net income (loss) (and the related per share measure) and its line item components certain items, as detailed in the reconciliation tables that follow, and in the case of non-GAAP adjusted net income (and the related per share measure), adjust for the income tax effect of non-GAAP adjustments and impact of the change in the statutory tax rate in the U.K. In this regard, the components of non-GAAP adjusted net income, including non-GAAP cost of product sales, non-GAAP SG&A expenses and non-GAAP R&D expenses, are income statement line items prepared on the same basis as, and therefore components of, the overall non-GAAP adjusted net income measure.
The Company believes that each of these non-GAAP financial measures provides useful supplementary information to, and facilitates additional analysis by, investors and analysts. In particular, the Company believes that each of these non-GAAP financial measures, when considered together with the Company’s financial information prepared in accordance with GAAP, can enhance investors’ and analysts’ ability to meaningfully compare the Company’s results from period to period and to its forward-looking guidance, and to identify operating trends in the Company’s business. In addition, these non-GAAP financial measures are regularly used by investors and analysts to model and track the Company’s financial performance. Jazz Pharmaceuticals’ management also regularly uses these non-GAAP financial measures internally to understand, manage and evaluate the Company’s business and to make operating decisions, and compensation of executives is based in part on certain of these non-GAAP financial measures. Because these non-GAAP financial measures are important internal measurements for Jazz Pharmaceuticals’ management, the Company also believes that these non-GAAP financial measures are useful to investors and analysts since these measures allow for greater transparency with respect to key financial metrics the Company uses in assessing its own operating performance and making operating decisions.
These non-GAAP financial measures are not meant to be considered in isolation or as a substitute for comparable GAAP measures; should be read in conjunction with the Company’s consolidated financial statements prepared in accordance with GAAP; have no standardized meaning prescribed by GAAP; and are not prepared under any comprehensive set of accounting rules or principles. In addition, from time to time in the future there may be other items that the Company may exclude for purposes of its non-GAAP financial measures; and the Company has ceased, and may in the future cease, to exclude items that it has historically excluded for purposes of its non-GAAP financial measures. For example, commencing in 2020, the Company no longer excludes upfront and milestone payments from the Company’s non-GAAP adjusted net income, its line item components and non-GAAP adjusted net income per diluted share. Likewise, the Company may determine to modify the nature of its adjustments to arrive at its non-GAAP financial measures. Because of the non-standardized definitions of non-GAAP financial measures, the non-GAAP financial measures as used by Jazz Pharmaceuticals in this press release and the accompanying tables have limits in their usefulness to investors and may be calculated differently from, and therefore may not be directly comparable to, similarly titled measures used by other companies.