
Aurora Cannabis Inc. Announces Results for the Fourth Quarter and 2018 Fiscal Year
- 223% Top Line Annual Revenue Growth
- Well Positioned for Canadian Adult Consumer Use Market
- Process Commenced to List Securities on Senior U.S. Exchange
EDMONTON, Sept. 24, 2018 /CNW/ – Aurora Cannabis Inc. (“Aurora” or the “Company”) (TSX: ACB) (OTCQB: ACBFF) (Frankfurt: 21P; WKN: A1C4WM), announced today its financial and operational results for the fourth quarter and fiscal year ended June 30, 2018.
Q4 2018 Financial and Operational Highlights
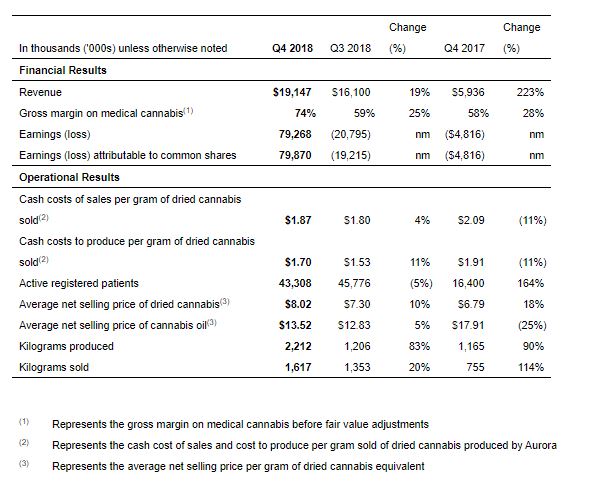 Q4 2018 Highlights:
Q4 2018 Highlights:
- Revenue of $19,147, up 223% compared to the same period in 2017.
- Pro-forma Q4 2018 revenue of $33,117, including MedReleaf
- Gross margin on medical cannabis of 74%, up 28% compared to the fourth quarter of 2017. This increase was primarily due to a higher average selling price per gram of dried cannabis, coupled with a higher proportion of cannabis oil sales in the Company’s sales ratio.
- Cash cost of sales and cash cost to produce per gram of dried cannabis sold both decreased 11% compared to the same period in 2017. This decrease was primarily due to efficiencies from automation and yield expertise.
- Active registered patients of 43,308, up 164% compared to the fourth quarter of 2017.
- Kilograms produced and kilograms sold of 2,212 and 1,617, up 90% and 114% respectively compared to the same period in 2017.
- 7 facilities with production licenses, 5 facilities with sales licenses
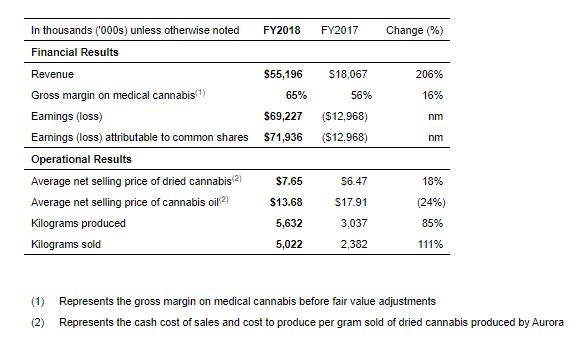 Fiscal 2018 Highlights:
Fiscal 2018 Highlights:
- Revenue of $55,196, up 206% compared to the prior year.
- Gross margin on medical cannabis of 65%, up 16% compared to FY 2017. This increase was primarily due to a higher average selling price per gram of dried cannabis, coupled with a higher proportion of cannabis oil sales in the Company’s sales ratio.
- Kilograms produced and kilograms sold of 5,632 and 5,022, up 85% and 111% respectively compared to FY 2017.
- 11 strategic acquisitions completed and 1 in progress.
- Total headcount grew to 1,400 from approximately 300 at the end of FY 2017.
- Pro-forma cannabis inventory and biological assets, including MedReleaf, of $88.8 million.
A comprehensive discussion of Aurora’s financials and operations are provided in the Company’s Management’s Discussion & Analysis (“MD&A”) and Financial Statements which will be filed on SEDAR at www.sedar.com and on the Aurora website.
Management Commentary
“Aurora made substantial progress toward our strategic goal of becoming the global scale and margin leader in the cannabis industry, establishing a vertically integrated company with a broadly diversified product offering with a large global footprint,” said Terry Booth, CEO of Aurora. “Our high-pace, consistent execution has enabled us to complete a number of transformative acquisitions, bringing together industry-leading companies in terms of scale, quality, efficiencies, plant and medical science, product development and innovation, brands, and international distribution.”
Mr. Booth added, “With coast-to-coast supply arrangements, and our strategic investment in Alcanna, we are very well positioned to capitalize on the significant adult consumer use opportunity in Canada. With reported Q4 revenues of $19.1 million, pro-forma Q4 revenues of over $33.1 million, and production capacity scaling up rapidly, we anticipate accelerated revenue growth during fiscal 2019. We have invested heavily in our organizational capabilities, including sales, marketing, and corporate talent and capacity, to ensure we will continue to drive strong and sustainable long-term growth.”
Today, Aurora ticks all the boxes for sustainable success and leadership in the cannabis industry. Aurora is capitalizing on a once in a lifetime opportunity, and establishing new industry standards in terms of execution, science, cultivation, international expansion and product development that position us exceptionally well to do so.
 Terry Booth, CEO of Aurora
Terry Booth, CEO of Aurora
We are very proud of our achievements over the past year, and we look forward to fiscal 2019 as we continue to execute.
U.S. Listing
Aurora intends to list its securities on a senior U.S stock exchange. In advance of a listing on a senior U.S. exchange, Aurora will file a Form 40-F Registration Statement with the United States Securities and Exchange Commission. The listing of the Company’s securities remains subject to exchange approval and the satisfaction of all applicable listing and regulatory requirements. A trading date will be made public once all regulatory formalities are satisfied.
Terry Booth, Aurora’s CEO added: “Listing our shares on a senior U.S. exchange reflects the level of corporate and business maturity and our high-paced execution. This listing provides access to a broader investor audience who gain the opportunity to participate in our continued success.”
Operational Highlights Subsequent to June 30, 2018
Facilities and Production update
During and subsequent to the quarter, the Company made significant progress towards increasing its production capacity. As at September 2018, the annualized run rate of Aurora’s in-service production rooms is 45,000 kgs. Management anticipates that around calendar year end 2018, the Company will have a production run rate in excess of 150,000 kg per annum, with subsequent scale up to over 500,000 kg per annum.
Aurora Sky
Aurora has completed full cycles in its first two licensed grow rooms and its mother room, and has submitted for final inspection towards obtaining its sales permit for Aurora Sky. The Company is pleased to announce that Health Canada has licensed two further production bays, each with more canopy space than the entire Aurora Mountain facility. Aurora has commenced populating these additional bays, bringing the total number of bays now in production (including mother room) to 5 of 17. The Company is in an ongoing process of obtaining production licenses as new bays come online, and anticipates submitting the license applications for the final rooms in November. Minor construction and commissioning items remain, which will be completed in advance of new bays coming online. Consequently, the Company continues on schedule towards reaching full 100,000 kg per annum production capacity around the end of calendar 2018.
Aurora Mountain
Continuous process improvement programs have resulted in an increase in production at Aurora Mountain, one of the Company’s EU GMP certified facilities. Management anticipates production at Mountain to reach 6,000 kg per annum, up by 25% from the facility’s previously disclosed capacity. The increase in production capacity at Mountain ensures the Company will be able to allocate more product to the supply restrained international markets.
Aurora Vie
The Company received its sales license for Aurora Vie in July 2018. The facility is now at full capacity, producing at a run rate of 4,000 kg per annum.
In Q4 2018, the Company completed the installation of Capcium softgel manufacturing equipment at Aurora Vie. The production line has been commissioned successfully, and the facility is currently producing high volumes of softgels for the adult consumer-use market to fulfill with orders received from provincial buyers.
Aurora Eau
Aurora’s newest facility received its production license in September 2018 and is now ramping up to full capacity at 4,500 kg per annum. Aurora Eau was purpose-built to EU GMP standards and represents the next evolution of Aurora’s indoor-grow facilities, where novel and exotic strains will be grown for both the medical and adult consumer-use markets.
Aurora Nordic, Phase I
Phase I of Aurora Nordic, the Company’s Danish facility, has been populated with genetics propagated from Aurora Mountain. The facility will ramp up to its full 8,000 kg per annum capacity in the coming months, increasing product availability for the EU markets.
MedReleaf Bradford
The MedReleaf Bradford facility is ramping up to full production capacity of 28,000 kg per annum. Currently, the facility is producing at a rate of approximately 10,000 kg per annum. The Bradford facility has large-scale oil production capacity, which will fuel the Company’s ability to produce higher margin products.
Aurora Sun
On April 16, 2018, the Company acquired approximately 71 acres of land in Medicine Hat, Alberta, for the construction of “Aurora Sun”, a highly automated cannabis production facility with ultra-low operating costs. The new facility will be 1.2 million square feet, 50% larger than Aurora’s Sky, a 100,000+ kg per annum Health Canada licensed facility at the Edmonton International Airport. Construction of Aurora Sun is on schedule with site preparation works completed and foundation work commenced.
Product Licensing
In addition to the licensing developments described above, Aurora reports it has received the following product and development licenses, broadening the Company’s higher-margin product offering, while facilitating research and exports:
- Capsule Licenses Granted: On July 3, 2018, Aurora’s wholly owned subsidiary, CanniMed, received Health Canada approval to commence sales of CanniMed Capsules, a line of vegan capsules which became available to patients on August 22, 2018.
- Aurora Mountain Dealer’s License: On July 30, 2018, Aurora obtained a Health Canada Dealer’s License under the Controlled Drugs and Substances Act for its EU GMP certified Aurora Mountain facility in Alberta. The new license provides Aurora additional opportunities to produce, assemble, and sell cannabis oils and future novel, derivative products from Aurora Mountain as well as import and export cannabis products to and from international markets, subject to applicable regulations.
- Softgel Capsules: On August 22, 2018, Aurora received Health Canada authorization to produce cannabis softgel capsules at its state-of-the-art Aurora Vie facility in Pointe-Claire, Québec. Immediately following the approval, Aurora started production of softgel capsules in partnership with Capcium Inc.
Canadian Adult Consumer-Use Market
In preparation for the commencement of legalized adult consumer use sales in Canada, from October 17, 2018 onwards, the Company has entered into supply arrangements with provinces and territories accounting for over 98% of the Canadian population. Consumer brands sold by Aurora, its subsidiaries and strategic partners will be available in Ontario, Québec, Alberta, British Columbia, Saskatchewan, Manitoba, Nova Scotia, the Yukon, North West Territories, Prince Edward Island, and Newfoundland and Labrador. During and subsequent to the quarter, the Company has been able to significantly increase its inventory. With production from the Company’s licensed facilities ramping up to full capacity, Aurora anticipates producing at a rate in excess of 150,000 kg per annum by the end of Q2 2019.
Alcanna Inc. (“Alcanna”)
In accordance with our strategic investment in Alcanna, the largest private sector liquor retailer in Canada, Alcanna intends to open 37 retail cannabis stores in Alberta (the maximum number permissible under AB regulations for year one), and is targeting additional locations across the country where private retail of cannabis is permissible. Alcanna, in preparation for the onset of private retail in Ontario from April 1, 2019 onwards, is in the process of securing a large number of locations for potential stores in the Ontario, if permitted under the regulatory framework, which is yet to be announced.
Acquisitions
The Company continued to execute on its strategy to build a global scale, science and margin leader. To this end, a number of acquisitions were completed, further increasing production capacity, geographic reach, science-driven product and technology development, and brand strength. Subsequent to the end of Q4 2018, Aurora acquired:
- MedReleaf Corp. (“MedReleaf”): Aurora completed the world’s largest cannabis industry transaction whereby Aurora acquired MedReleaf. The transaction has created a unified cannabis industry leader with a combined funded capacity of more than 500,000 kg per annum, broad domestic and international distribution channels, industry-leading product development capabilities and strong medical and consumer brands.
- HotHouse Consulting Inc. (“HotHouse”): On August 7, 2018, the Company entered into a Letter of Intent to acquire HotHouse, a greenhouse consulting business with an emphasis on large scale cannabis production.
- Anandia Laboratories Inc. (“Anandia”): On August 8, 2018, the Company acquired Anandia, a global leader in cannabis science (genetics, breeding) and analytical product testing. The transaction enables the Company to develop new strains with specific terpene/cannabinoid profiles for targeted product applications, as well as strains with improved cultivation characteristics. Management believes these activities will lead both to the development of new, higher-margin products and a further increase in efficiency of its cultivation processes.
- ICC Labs Inc. (“ICC”): On September 10, 2018, Aurora entered into an agreement to acquire ICC for an aggregate purchase price of $290 million. ICC, the leading cannabis company in South America, adds significant low-cost production and processing capacity of both THC and CBD based products in both Uruguay and Colombia. In Uruguay, the world’s first country to legalize adult consumer use of cannabis, ICC maintains 70% market share and a broad and well-diversified product portfolio, as well as extensive distribution channels throughout South America and internationally.
- Agropro UAB (“Agropro”) and Borela UAB (“Borela”): On September 12, 2018, Aurora acquired Europe’s largest producer, processor and supplier of certified organic hemp and hemp products, Agropro, as well as hemp processor and distributor Borela. This acquisition is anticipated to yield significant quantities of CBD for extraction, and is expected to create further synergies through the Company’s CBD and hemp product value chain, which includes majority ownership of Hempco Food and Fiber.
Integration
The Company has developed the integration of acquisitions into a core competency. Under the leadership of Andre Jerome, SVP, Business Integrations, who formerly implemented global business integrations for Vodafone, a $65 billion telecommunications company, the Company has developed a methodology that ensures rapid integration of acquired companies into the Aurora organization. Integration of CanniMed was fully completed within the targeted 90-day timeline, and has resulted in an acceleration of its development, including a significant improvement in production rates of dried cannabis and cannabis oils.
Integration of MedReleaf into the Aurora organization has commenced and is progressing according to schedule. Mutual learning and implementation of best practices is a core aspect of the MedReleaf integration, and teams at both companies have identified key performance indicators, based on which the integration program is being executed.
Strategic Investments & Agreements
During and subsequent to the quarter, the Company made a number of strategic investments in both public and private companies. As at June 30, 2018, the unrealized gains on its public investments made to date stood at $372.0 million. Significant strategic investments made subsequent to June 30, 2018 consist of:
- Evio Beauty Group Ltd. (“Evio”): On July 10, 2018, Aurora entered into a Product Development and Distribution Agreement with Evio to develop and manufacture a line of co-branded topical cosmetic products formulated with a cannabinoid or cannabinoids.
- CannaRoyalty Corp. (“CannaRoyalty”): On August 1, 2018, the Company entered into an agreement with CannaRoyalty whereby CannaRoyalty assigned to Aurora all of its right, title, and interest in an exclusive license for a technology for creating machine-rolled cannabis developed by Wagner Dimas Inc. (“Wagner”). The Wagner technology has now been installed at Aurora, and the large-scale production of pre-rolled product has commenced in preparation to filling orders received from provincial buyers who will be supplying the adult consumer user market.
International Developments
Aurora continues to expand its international footprint and capitalize on its early mover advantage in key growth markets:
- Malta: Aurora entered into a Letter of Intent with Malta Enterprises concerning the establishment of the first seed-to-pharma cannabis facility in Malta, subject to certain conditions. The Company anticipates the facility, to be designed by Aurora Larssen Projects, to be focused on the production of higher margin derivative products, aimed at the Maltese and Southern European markets.
- EU GMP Certification: Aurora’s wholly-owned subsidiary MedReleaf received full EU GMP certification for its Markham facility. The certification of the Markham facility will increase product availability for the rapidly growing, higher-margin and heavily regulated EU market. All of the Company’s facilities are being designed and built to EU GMP standards, including Aurora Nordic Phase I, which currently is in production and ramping up to full capacity.
- Establishing Aurora Europe: Aurora established a pan-European company, Aurora Europe GmbH, headquartered in Berlin, Germany. Pedanios GmbH, Europe’s largest distributor of cannabis, now operates as Aurora Deutschland GmbH, while the Company has also formed Aurora Italia, Aurora Nordic (Denmark) and a number of other local companies.
- Australis Capital Inc. (“ACI”): On September 19, 2018, the Company completed the spin-out of ACI, an independent company, and distributed to Aurora shareholders, as a return of capital, units of ACI on the basis of one unit for every 34 Aurora shares. The units commenced trading on the CSE on September 19, 2018. ACI is an investment company with a focus on the U.S. cannabis market, which is characterized by large fragmentation and limited access to capital. ACI’s management, board and advisory teams have deep experience and relationships within the cannabis industry, and believe they will be able to secure investments to build significant shareholder value.
- Cayman Islands: sales of CanniMed oils to the Cayman Islands continue to grow in line with patient demand. The Company signed a supply agreement
International hemp: Through the acquisitions of Agropro and Borela, as well as the planned acquisition of ICC, the Company is well positioned to capitalize on the rapidly growing hemp-based food product markets. Agropro and Borela products are currently available in 27 countries. Going forward, the Company intends to create further integration with its majority owned Hempco Food and Fiber, further increasing market reach and product development capabilities. - MED Colombia: through the acquisition of MedReleaf, the Company now owns MED Colombia, a licensed cannabis company in Colombia with substantial grow potential and a strong portfolio of genetics. Upon successful completion of the ICC acquisition, MED Colombia will become part of Aurora’s South American platform.
- Australia: Aurora recently exported oil products to Australia, which were supplied to patients through its partially-owned strategic partner Cann Group. Cann Group has announced it will be constructing an ALPS (Aurora Larssen Projects) designed high-technology, hybrid cultivation facility at the Melbourne International Airport. Aurora and its wholly-owned subsidiary Anandia have also successfully exported plant tissue culture derived genetics for Cann Group to enhance its cultivation program.
Financing
- Bank of Montreal (“BMO”) Debt Facility: the Company finalized a $200,000 Debt Facility (the “Facility”) with BMO consisting of a $150,000 term loan and a $50,000 revolving credit facility, both of which will mature in 2021. The Company also has the option to upsize the Facility to a total of $250,000, subject to certain conditions. The Facility will be primarily secured by Aurora’s production facilities and can be repaid without penalty at Aurora’s discretion. The Facility with BMO is the largest of its kind in the cannabis industry with a tier one bank and a validation, management believes, of the quality and economic value of Aurora’s facilities and of the Company’s prospects.
Q4 2018 Operational Highlights
Strategic Investments
- Hempco Food and Fiber Inc. (Hempco): The Company purchased an additional 10.8 million shares to increase its ownership interest to 52.3%. Hempco plays an important role in the Company’s strategy to secure access to low-cost material for the potential production of CBD extracts.
- CTT Pharmaceuticals Inc. (“CTT”): The Company acquired a 9.41% interest in CTT, who have developed fast-dissolving oral thin wafers that provide dose-specific, smoke-free delivery of medical cannabis with a rapid onset of action. The companies are jointly working towards approval by Health Canada for market introduction.
- Choom Holdings Inc. (“Choom”): The Company subscribed to an investment representing an 8% ownership interest in Choom, extending the Company exposure to the emerging craft cultivation market, and expanding its reach into the cannabis retail market.
- Capcium Inc. (“Capcium”): The Company acquired a 19.99% ownership interest in Capcium, a leading softgel manufacturer in the cannabis industry. The Capcium technology has been installed at Aurora Vie and the Company has commenced the large-scale production of softgels in response to orders from provincial buyers to service the Canadian adult consumer-use market.
- Green Organic Dutchman Holdings Ltd. (“TGOD”): The Company holds approximately 17% of TGOD. In addition to securing 20% of the output of TGOD’s Ancaster and Valleyfield facilities, the latter of which is constructed with the assistance from Aurora Larssen Projects, the value of the Company’s investment has appreciated significantly. The Company has options to increase its ownership in TGOD to over 50%.
Supply Agreements and Partnerships
- Pharmasave: On April 4, 2018, CanniMed, a wholly-owned subsidiary of Aurora, entered into a Letter of Intent with Pharmasave, a network of over 650 independently-owned pharmacies, to become a preferred supplier of medical cannabis.
Société des Alcools du Québec (“SAQ”)”: On April 11, 2018, the company completed a final agreement with SAQ to supply cannabis for the Quebec consumer market, once legalized.
Acquisitions - CanniMed Therapeutics Inc. (“CanniMed”): On May 1, 2018, the Company completed the acquisition of CanniMed. The transaction creates strong strategic synergies, in particular for the domestic and international medical cannabis markets, in terms of distribution, product development, and branding. Integration of CanniMed into Aurora is complete and acceleration of CanniMed’s production and other operations has commenced.
International Developments
- Italy: Aurora completed the successful delivery of the first ever batch of privately exported medical cannabis from Canada to the Italian government through its wholly-owned German subsidiary Aurora Deutschland GmbH (“Aurora Deutschland”, formerly Pedanios GmbH)
- Germany: Aurora, through Aurora Deutschland, signed a collaboration agreement with Heinrich Klenk GmbH & Co. KG (“Klenk”), one of Europe’s largest medicinal plant companies. Klenk’s products are carried in over 25,000 pharmacies throughout Germany and Europe. Under the terms of the agreement, Aurora launched a new cannabis brand in Germany called “Cannabis Klenk” which is produced in Canada, imported by Aurora Deutschland, and sold to German pharmacies through Klenk’s existing and wide-reaching pharmaceutical wholesale distribution network.
- Malta: Aurora, through its wholly-owned European subsidiary Aurora Deutschland, became the first licensed supplier of medical cannabis to patients in Malta, the third country in the European Union where Aurora Deutschland currently sells medical cannabis.
Outlook
Aurora is exceptionally well‑positioned in all of its markets, including adult consumer-use market, Canadian medical and the international medical markets, with compelling brands and strong patient and consumer recognition.
In fiscal 2019, the Company will continue to focus on expanding capacity and sales growth in all its markets, in addition to further product development, continued international expansion and realization of acquisition synergies.
Aurora is rapidly accelerating production out of its newly licensed facilities, starting with Aurora Sky, which is expected to ramp to full 100,000 kg per annum capacity around calendar year end 2018. The Company anticipates to reach a production run rate of approximately 150,000 kg per annum around year end, scaling up subsequently to over 500,000 kg per annum through further “Sky Class” facilities, Aurora Sun and Aurora Nordic. The high degree of automation, and customized and fully controlled growing conditions at the Sky Class facilities are anticipated to result in production costs well below one dollar per gram. Management believes these factors together will deliver high growth and continuously improving margins.
While the historic milestone of Canada becoming the first G7 nation to legalize the adult consumer-use market creates a very significant growth opportunity, the Company maintains its position that the international medical has the most significant growth prospects, and is expected to grow to 10 million kilograms per annum. The Company has established significant early mover advantage, has a presence on five continents, and is Europe’s largest distributor of medical cannabis. Aurora also owns two of only six facilities in the world that are EU GMP certified, ensuring continued access to restrictive markets. This early mover advantage, management believes, will enable the Company to establish significant market share in the global medical market.
Financial review Q4 2018
Revenue
In the fourth quarter of 2018 (“Q4 2018”) cannabis revenue grew to $14.9 million, a 38% increase compared to the third quarter of 2018 (“Q3 2018”) and a 113% increase compared to the fourth quarter of 2017 (“Q4 2017”). Total revenue grew to $19.1 million, representing a 19% increase compared to Q3 2018 and a 223% increase compared to Q4 2017. Revenue growth compared to the same quarter in the prior year was attributable mainly to higher patient numbers following the acquisition of CanniMed, increased product availability through scale up of operations and the CanniMed acquisition, an increase in the average net selling price of dried cannabis, development of international markets, and product diversification.
Average price of product sold was $9.20 per gram in Q4 2018, an increase of 15% compared to Q3 2018 and 23% compared to Q4 2017 as a result of an increase in cannabis oils sold. Total product sold was 1,617 kilograms of dried cannabis and cannabis oils in Q4 2018, an increase of 19% as compared to Q3 2018, and 114% compared to Q4 2017.
Total cannabis inventory and biological assets increased 248% to $41.0 million in Q4 2018 compared to Q4 2017 as Aurora chose to constrain international sales in order to continue servicing the Canadian medical market, while building inventory in preparation for the Canadian adult consumer-use market. This onetime constraint on international sales has been alleviated by the rapid completion of Aurora’s Canadian production facilities (including Sky, Vie & Eau), as well as the EU GMP certification of MedReleaf’s Markham facility, and ongoing yield improvements at CanniMed.
Operating Expenses
Throughout 2018, Aurora continued to make significant investments in its infrastructure and talent, scaling the organization to better realize the tremendous opportunity ahead in the domestic and international medical cannabis markets, and the upcoming Canadian adult consumer-use market.
As a result, general and administration costs (“G&A”) increased to $22.6 million in Q4 2018, compared to $9.8 million in Q3 2018. Sales and marketing costs (S&M) in Q4 2018 increased to $14.8 million dollars, from $5.9 million in Q3 2018, as a result of investments in our overall brand strategy which included certain one-time activities in preparation for the impending adult consumer-use recreational market in Canada. The integration of CanniMed accounted for 25% of the increase to G&A and 19% of the increase to S&M.
Cost of sales
Cash cost of sales per gram of dried cannabis sold and cash cost to produce per gram of dried cannabis sold increased by $0.07 and $0.17 respectively from the prior quarter, mainly due to the inclusion of CanniMed’s higher per unit production costs, partially offset by lower utility costs in the summer months. Aurora continues to drive yield and efficiency improvements at CanniMed in line with Aurora’s other operating facilities, and anticipates production costs to come down further.
Gross Profit
Q4 2018 gross profit was $20.6 million, compared to a $5.8 million in Q4 2017. The increase in gross profit during the period was partially attributable to the net effect of changes in fair value of biological assets and a decrease in the cost of sales for medical cannabis on a per gram basis.
Net Income
Q4 2018 net income increased to $79.3 million, compared to a net loss of $20.8 million in Q3 2018 and $4.8 million in Q4 2017. The increase was primarily attributable to the unrealized non-cash gain on derivatives and marketable securities, which was partially offset by increased finance costs, share-based payments, acquisition and project evaluation costs.
Cash Position, Cash Flows, and Working Capital
Net cash and cash equivalents on hand decreased from $159.7 million at the end of Q4 2017 to $89.2 million as at Q4 2018. Non-cash working capital at the end of Q4 2018 was $56.4 million, as compared to $10.4 million at the end of Q4 2017. The change in working capital was largely attributable to an increase in marketable securities and the planned buildup of inventory, partially offset by accounts payable and accrued liabilities related to construction of our production facilities. Additionally, as at September 21, 2018, the market value of the Company’s investments in public companies exceeded $700 million.
The Company anticipates that it has sufficient liquidity and capital resources to meet all of its currently planned expenditures for the next twelve months.
Pro-Forma Reconciliation
For the three months ended June 30, 2018, pro-forma revenue, including the results of MedReleaf, would have been $33.1 million. As at June 30, 2018, cash and cash equivalents would have been $255.2 million and cannabis inventory and biological assets would have been $88.8 million.
About Aurora
Headquartered in Edmonton, Alberta, Canada with funded capacity in excess of 500,000 kg per annum and sales and operations in 18 countries across five continents, Aurora is one of the world’s largest and leading cannabis companies. Aurora is vertically integrated and horizontally diversified across every key segment of the value chain, from facility engineering and design to cannabis breeding and genetics research, cannabis and hemp production, derivatives, high value-add product development, home cultivation, wholesale and retail distribution.
Highly differentiated from its peers, Aurora has established a uniquely advanced, consistent and efficient production strategy, based on purpose-built facilities that integrate leading-edge technologies across all processes, defined by extensive automation and customization, resulting in the massive scale production of high quality product at ultra-low costs. Intended to be replicable and scalable globally, these production facilities are designed to produce cannabis of significant scale, with high quality, industry-leading yields, and ultra-low per gram production costs. Each of Aurora’s facilities is built to meet EU GMP standards, and its first production facility, the recently acquired MedReleaf Markham facility, and its wholly owned European medical cannabis distributor Aurora Deutschland (formerly Pedanios), have achieved this level of certification.
In addition to the Company’s rapid organic growth and strong execution on strategic M&A, which to date includes 15 companies – MedReleaf, CanvasRX, Peloton Pharmaceutical, Aurora Deutschland (formerly Pedanios), H2 Biopharma, Urban Cultivator, BC Northern Lights, Larssen Greenhouses, CanniMed Therapeutics, Anandia Labs, HotHouse Consulting, Agropro, Borela, and the pending acquisition of ICC Labs – Aurora is distinguished by its reputation as a partner and employer of choice in the global cannabis sector, having invested in and established strategic partnerships with a range of leading innovators, including: The Green Organic Dutchman Holdings Ltd. (TSX: TGOD), Radient Technologies Inc. (TSXV: RTI), Hempco Food and Fiber Inc. (TSXV: HEMP), Cann Group Ltd. (ASX: CAN), Micron Waste Technologies Inc. (CSE: MWM), Choom Holdings Inc. (CSE: CHOO), Namaste Technologies Inc. (TSXV: N), Evio Beauty Group (private), Wagner Dimas (private), CTT Pharmaceuticals (OTCC: CTTH), and Alcanna Inc. (TSX: CLIQ).
Aurora’s Common Shares trade on the TSX under the symbol “ACB”, and are a constituent of the S&P/TSX Composite Index.
For more information about Aurora, please visit our investor website, investor.auroramj.com, Twitter, Facebook or Instagram
