GW Pharmaceuticals plc Reports Fourth Quarter and Year-End 2019 Financial Results and Operational Progress
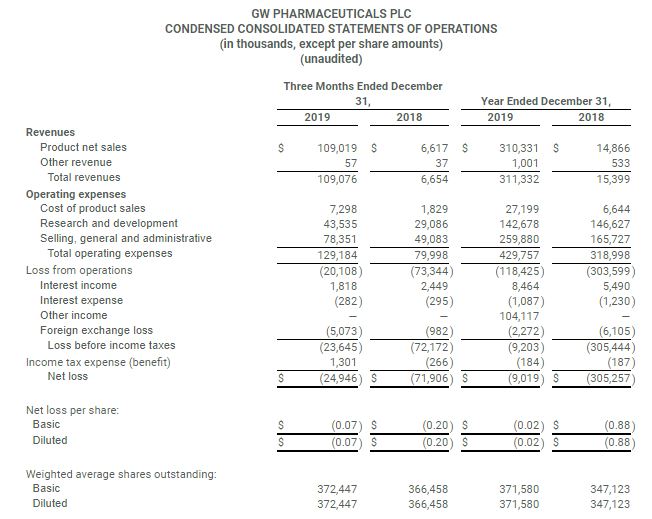
- Total revenue of $109.1 million for the fourth quarter and $311.3 million for the full year
- Total Epidiolex® net product sales of $104.5 million for the fourth quarter and $296.4 million for the first full year of sales
- Conference call today at 4:30 p.m. EST
LONDON and CARLSBAD, Calif., Feb. 25, 2020 (GLOBE NEWSWIRE) — GW Pharmaceuticals plc (Nasdaq: GWPH), a world leader in the science, development, and commercialization of cannabinoid prescription medicines, today announces financial results for the fourth quarter and full-year ended December 31, 2019.
2019 was an exceptional and transformative year for GW, led by the successful launch of Epidiolex in the US and approval in Europe. The positive impact this medicine has had on thousands of patients and their families provides a compelling foundation for continued growth in 2020.
 Justin Gover, GW’s Chief Executive Officer
Justin Gover, GW’s Chief Executive Officer
We also expect 2020 to be an important year for our growing and developing product pipeline beyond Epidiolex as we build on our world leadership in cannabinoid science. We are focused on advancing nabiximols in the US in several indications and clinical programs with other potential products whilst continuing to bring Epidiolex to more patients in the US and Europe.
OPERATIONAL HIGHLIGHTS
- Significant progress with Epidiolex (cannabidiol)
- Total net product sales of Epidiolex of $104.5 million for the fourth quarter and $296.4 million for the full year
- U.S. commercial update
- 2020 focus on broadening prescriber base, expanding payer coverage, entering long term care segment, and expected launch of TSC indication
- European launch underway
- German commercial launch in Q4 2019
- Secured positive NICE recommendation in the UK with commercial launch in Q1 2020
- Commercial launches in France, Spain and Italy expected later this year, following pricing and reimbursement
- Clinical progress with further indications – broadening addressable market
- Tuberous Sclerosis Complex (TSC) sNDA filed with FDA earlier this month and MAA submission to EMA expected in Q1 2020
- Phase 3 trial in Rett Syndrome recruiting
- Several new formulations of CBD advancing into additional Phase 1 studies in 2020, including modified oral solution, and capsule
- Improved intellectual property rights and exclusivity
- In addition to orphan exclusivity, 9 granted patents listed in “Orange Book” and align directly with Epidiolex FDA label with expiry dates to 2035
- Epidiolex composition patent application recently published
- 2 new allowed patents broadly covering use in LGS and Dravet syndrome
- Additional patent applications under review, including patents related to the use of Epidiolex in TSC and other indications
- Pipeline progress
- Nabiximols (Sativex® outside of the US)
- Multiple Sclerosis spasticity -3 positive Phase 3 trials completed in Europe
- US pivotal clinical program expected to commence in Q2 2020 to augment existing data package
- Spinal cord injury spasticity – clinical program expected to commence in H2 2020
- PTSD – clinical program expected to commence in H2 2020
- Multiple Sclerosis spasticity -3 positive Phase 3 trials completed in Europe
- Schizophrenia (GWP42003)
- Positive Phase 2a trial published and Phase 2b trial expected to commence H1 2020
- CBDV in autism
- 30-patient open label study in autism underway. Initial data expected in 2020.
- Investigator-led 100 patient placebo-controlled trial in autism underway
- Open label study in Rett syndrome and seizures underway
- Neonatal Hypoxic-Ischemic Encephalopathy (NHIE) intravenous CBD program
- Phase 1b safety study in patients underway
- Orphan Drug and Fast Track Designations granted from FDA and EMA
- Nabiximols (Sativex® outside of the US)
FINANCIAL HIGHLIGHTS
- Total revenue for the quarter ended December 31, 2019 was $109.1 million compared to $6.7 million for the quarter ended December 31, 2018
- Net loss for the quarter ended December 31, 2019 was $24.9 million compared to a net loss of $71.9 million for the quarter ended December 31, 2018
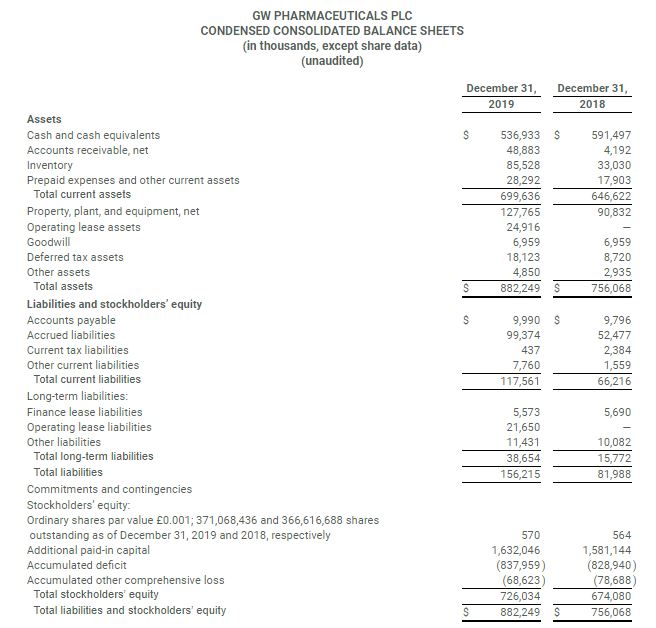
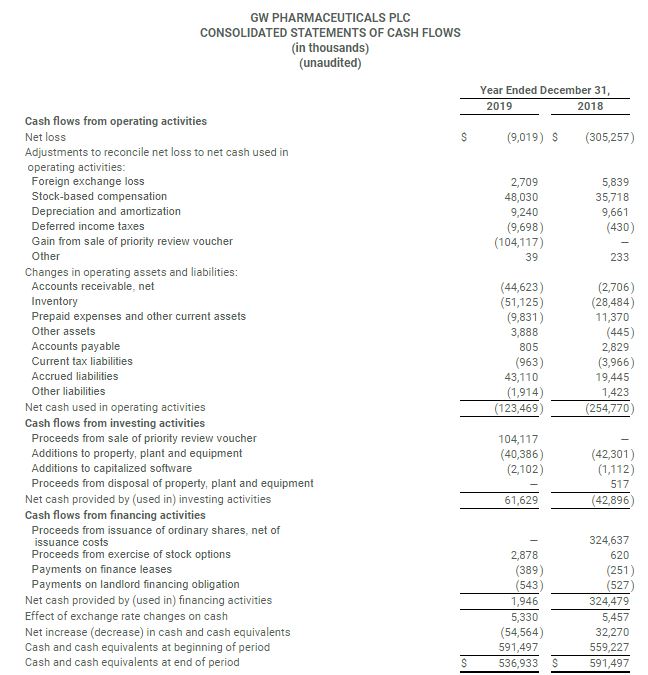
- Cash and cash equivalents at December 31, 2019 were $536.9 million compared to $591.5 million as of December 31, 2018
Conference Call and Webcast Information
GW Pharmaceuticals will host a conference call and webcast today at 4:30 pm EST. To participate in the conference call, please dial 877-407-8133 (toll free from the U.S. and Canada) or 201-689-8040 (international). Investors may also access a live audio webcast of the call via the investor relations section of the Company’s website at http://www.gwpharm.com. A replay of the call will also be available through the GW website shortly after the call and will remain available for 90 days. Replay Numbers: (toll free):1-877-481-4010 or 919-882-2331 (international). For both dial-in numbers please use conference Replay ID: 33178.
About GW Pharmaceuticals plc and Greenwich Biosciences, Inc.
Founded in 1998, GW is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. The Company’s lead product, EPIDIOLEX® (cannabidiol) oral solution CV, is commercialized in the U.S. by its U.S. subsidiary Greenwich Biosciences for the treatment of seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients two years of age or older. This product has received approval in the European Union under the tradename EPIDYOLEX®. The Company has submitted a supplemental New Drug Application to the U.S. Food and Drug Administration (FDA) to expand the indication for Epidiolex to include seizures associated with Tuberous Sclerosis Complex, for which it has reported positive Phase 3 data, and is carrying out a Phase 3 trial in Rett syndrome. The Company has a deep pipeline of additional cannabinoid product candidates, in particular nabiximols, for which the Company is advancing multiple late-stage clinical programs in order to seek FDA approval in the treatment of spasticity associated with multiple sclerosis and spinal cord injury, as well as for the treatment of PTSD. The Company has additional cannabinoid product candidates in Phase 2 trials for autism and schizophrenia. For further information, please visit www.gwpharm.com.