GW Pharmaceuticals plc Reports Fourth Quarter and Year-End 2020 Financial Results and Operational Progress
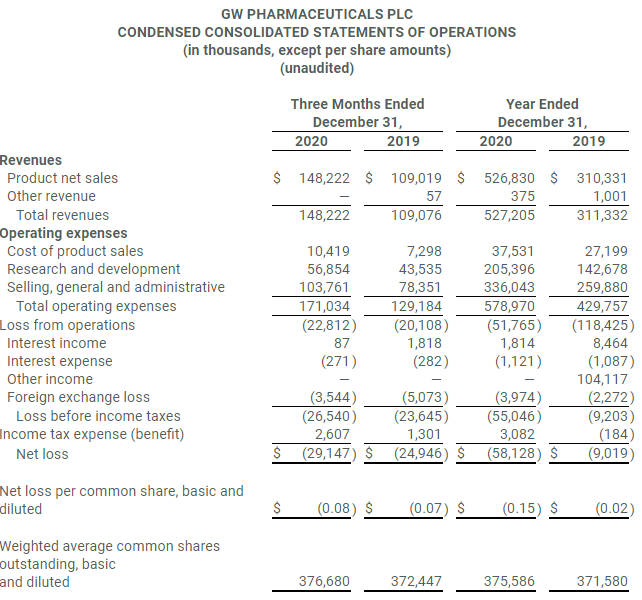
- Total revenue of $148.2 million for the fourth quarter and $527.2 million for the full year
- Total Epidiolex® net product sales of $144.1 million for the fourth quarter and $510.5 million for the full year
- Previously announced agreement to be acquired by Jazz Pharmaceuticals; transaction expected to close in Q2 2021
LONDON and CARLSBAD, Calif., Feb. 16, 2021 (GLOBE NEWSWIRE) — GW Pharmaceuticals plc (Nasdaq: GWPH), a world leader in the science, development, and commercialization of cannabinoid prescription medicines, today announced financial results and operating progress for the fourth quarter and full-year ended December 31, 2020.
We are very proud of our strong financial performance and operational progress in 2020, as Epidiolex sales increased by more than 70% during the year despite the challenges of COVID-19.
 Justin Gover, chief executive officer of GW
Justin Gover, chief executive officer of GW
We are well positioned to build on our success and continue to deliver strong growth in 2021 in both the U.S. and Europe, where we continue to make progress preparing for several commercial launches that are expected later this year.
He also added, “We have commenced our Phase 3 clinical program for nabiximols in the treatment of multiple sclerosis spasticity, which provides multiple opportunities for an NDA submission. Beyond nabiximols, we are advancing a diverse and robust neuroscience pipeline with several preclinical and clinical-stage pipeline candidates as part of our commitment to patients and to developing innovative medicines that address significant unmet needs. We have strong momentum and a tremendous opportunity to continue to build on our global cannabinoid leadership position as we prepare to join Jazz Pharmaceuticals and transform the lives of even more patients and families.”
FINANCIAL RESULTS
- Total revenue for the quarter ended December 31, 2020 was $148.2 million compared to $109.1 million for the quarter ended December 31, 2019.
- Total revenue for the full-year 2020 was $527.2 million, a 69 percent increase compared to $311.3 million for the prior year period.
- Net loss for the quarter ended December 31, 2020 was $29.1 million compared to net loss of $24.9 million for the quarter ended December 31, 2019.
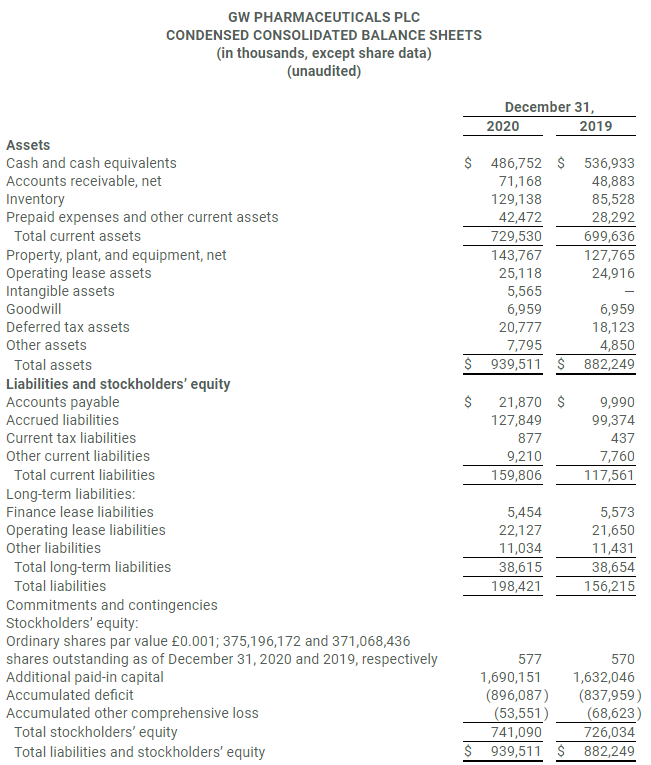
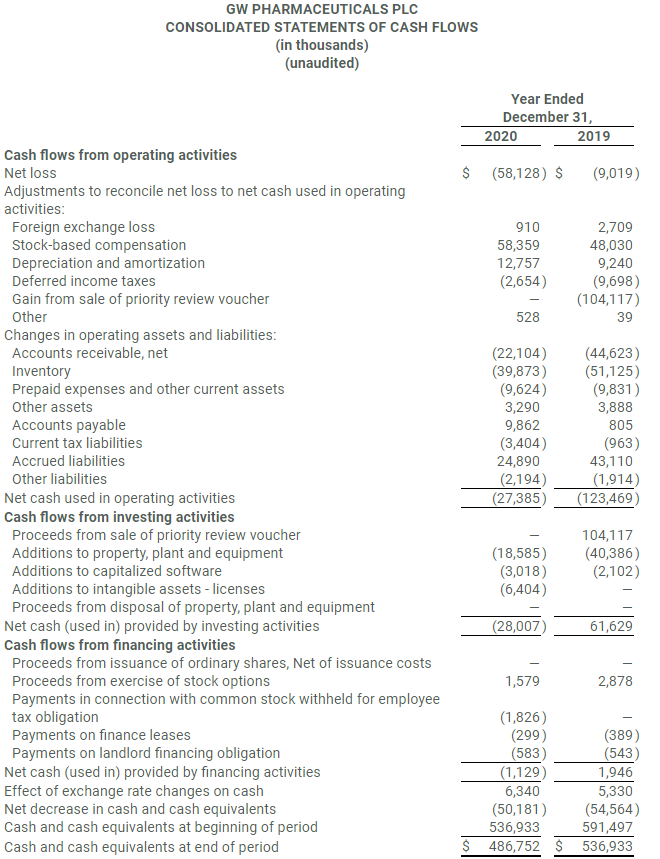
- Cash and cash equivalents at December 31, 2020 were $486.8 million.
OPERATIONAL HIGHLIGHTS
- Epidiolex (cannabidiol) progress:
- Total net product sales of Epidiolex of $144.1 million for the fourth quarter and $510.5 million for the year ended December 31, 2020.
- U.S. commercial update
- U.S. Epidiolex net product sales of $128.8 million for the fourth quarter and $467.6 million for the year ended December 31, 2020
- TSC indication launched with high prescriber awareness and near universal payer coverage
- Expanded payer coverage
- More than 110 million lives with no/broad prior authorization (70% increase in 2020)
- Ex-U.S. commercial update
- Ex-U.S. Epidyolex Q4 2020 net product sales of $15.3 million and full-year 2020 sales of $42.9 million
- Continued progress expanding global reach of Epidyolex:
- Pricing and reimbursement approved in Germany, Finland and Israel
- Swissmedic approval received for the adjunctive therapy of seizures associated with LGS and DS
- Launches in France, Spain and Italy expected in H1 2021
- EMA TSC approval expected H1 2021
- Strengthening commercial exclusivity
- Orphan exclusivity in both the U.S. and EU
- 14 patents listed in Orange Book, 13 of which expire in 2035
- Patents include formulation and method of use
- An additional patent has been granted and will be listed in the Orange Book in Q1 2021 and a further patent is expected to be granted and listed in the Orange Book in Q2 2021
- Epidiolex composition patent application filed
- Nabiximols development program:
- MS Spasticity trials underway
- Phase 3 placebo-controlled spasm frequency study (N=450)
- Phase 3 placebo-controlled muscle tone study (N=52)
- MS Spasticity trials due to commence
- Phase 3 placebo-controlled muscle tone studies:
- N=190; Expected start: Q2 2021
- N=36 (nabiximols responders); Expected start: Q2 2021
- Additional Phase 3 placebo-controlled spasm frequency study (N=200) in nabiximols responders expected start Q2 2021
- Phase 3 placebo-controlled muscle tone studies:
- Spinal Cord Injury (SCI) spasticity clinical program
- First SCI trial underway
- N=~100 observational clinical discovery study
- SCI spasticity trials due to commence
- N=~160 (muscle tone in nabiximols responders); Placebo-controlled parallel group design. Expected start: 2021
- N=~400 (spasm frequency); Placebo-controlled parallel group design. Expected start: 2021
- First SCI trial underway
- MS Spasticity trials underway
- Additional pipeline programs:
- Schizophrenia (GWP42003)
- Phase 2b trial now actively recruiting
- Autism:
- CBD formulation Phase 2 study expected to commence in Q1 2021
- CBDV investigator-led 100 patient placebo-controlled trial in autism underway
- New botanical cannabinoid pipeline product (GW541)
- Phase 1 trial underway
- Potential targets within field of neuropsychiatry
- Neonatal Hypoxic-Ischemic Encephalopathy (NHIE) intravenous CBD program
- Phase 1b safety study in patients continues to recruit
- Orphan Drug and Fast Track Designations granted from FDA and EMA
- Novel cannabinoid molecule synthesis and preclinical development
- At least one program expected to enter Phase 1 in 2021
- Several other molecules have demonstrated preclinical efficacy and are advancing towards the clinic
- Schizophrenia (GWP42003)
- On Feb. 3, 2021, Jazz Pharmaceuticals plc (Nasdaq: JAZZ) and GW announced the companies had entered into a definitive agreement for Jazz to acquire GW for $220.00 per American Depositary Share (ADS), in the form of $200.00 in cash and $20.00 in Jazz ordinary shares (subject to limitations on the maximum and minimum number of Jazz ordinary shares issuable per ADS), for a total consideration of $7.2 billion. The transaction is subject to the approval of GW shareholders, sanction by the High Court of Justice of England and Wales and other customary closing conditions, including regulatory approvals. Subject to the satisfaction or waiver of the closing conditions, the transaction is expected to close in the second quarter of 2021.
Conference Call/Earnings Materials
Given the recently announced agreement for GW to be acquired by Jazz Pharmaceuticals, GW will no longer hold conference calls. Earnings materials are available publicly on the Investor Relations page of GW’s website at http://www.gwpharm.com. Questions may be directed to Investor Relations via e-mail at the contact information below.
About GW Pharmaceuticals plc and Greenwich Biosciences, Inc.
Founded in 1998, GW is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. The company’s lead product, EPIDIOLEX® (cannabidiol) oral solution, is commercialized in the U.S. by its U.S. subsidiary Greenwich Biosciences for the treatment of seizures associated with Lennox-Gastaut syndrome (LGS), Dravet syndrome, or tuberous sclerosis complex (TSC) in patients one year of age and older. This product has received approval in the European Union under the tradename EPIDYOLEX® for the adjunctive treatment of seizures associated with LGS or Dravet syndrome in conjunction with clobazam in patients two years and older and is under EMA review for the treatment of TSC. The company has a deep pipeline of additional cannabinoid product candidates, in particular nabiximols, for which the company is advancing multiple late-stage clinical programs in order to seek FDA approval in the treatment of spasticity associated with multiple sclerosis and spinal cord injury. The company has additional cannabinoid product candidates in clinical trials for autism and schizophrenia.