 MediPharm Labs Reports Fourth Quarter and Full Year 2019 Results
MediPharm Labs Reports Fourth Quarter and Full Year 2019 Results
- 2019 Revenue $129 Million, Adjusted EBITDA1 $25 Million
- Q4 2019 Revenue $32 Million, Adjusted EBITDA1 $3 Million
- Year-end Cash and Equivalents of $39 million
BARRIE, Ontario, March 30, 2020 (GLOBE NEWSWIRE) — MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven pharmaceutical-quality cannabis extraction, distillation and derivative products today announced its financial results for the three and twelve months ended December 31, 2019 and reaffirmed its strategic priority to build a multi-jurisdictional, GMP-certified pharmaceutical-grade platform to serve the world’s most attractive emerging cannabis medical markets.
2019 was a transformational year for MediPharm Labs. We were one of only a very few cannabis companies to successfully execute on its strategy and be profitable.

Pat McCutcheon, Chief Executive Officer, MediPharm Labs
We established ourselves as a high-quality extract Company, earning our first GMP certification, positioning ourselves to become a global supplier of pharma-quality products as future brands, larger consumer packaged goods companies and pharmaceutical companies begin to enter the market.
“We move forward with a strengthened market leadership position and diversified platform. Our portfolio of licences and first licensed offshore facility in Australia position us well to create new and innovative products at home and abroad and to achieve global expansion creating tangible value for customers and shareholders.”
2019 BUSINESS HIGHLIGHTS AND RECENT ACCOMPLISHMENTS
- First full year of profitability and a top five cannabis revenue company in the Canadian Cannabis industry
- Signed multiple new supplier agreements with other Cannabis Act licensed holders, direct-to-consumer brands, provincial and private retailers including Medical Cannabis by Shoppers (2020); first international export agreements with AusCann Group Holdings Ltd., an Australian medicinal cannabis company, and ADREXpharma GmbH, a German pharmaceutical distributor
- Received first GMP certification by the Therapeutic Goods Administration of Australia (TGA) and completed its subsidiary’s offshore facility in Australia
- Successful ramp up of the Company’s operations to support customers in the development and introduction of a range of Cannabis 2.0 products; first shipments delivered prior to year-end
- Successful shipment of the Company’s first international exports of wholesale cannabis extract to Australia
- Achieved a Research Licence and conducted its first controlled human administration trials in Barrie
- Expanded licensed facility footprint, to 25,000 sq. ft. to scale up automated downstream production, packaging and R&D supporting fulfillment of new product formats and formulation orders in 2020
2019 FINANCIAL SUMMARY1,2
- Revenue of $129.3 million reflecting a leadership position serving multiple customers in the wholesale concentrate market in Canada as well as initial international wholesale exports to Australia
- Gross profit of $42.9 million, gross margin of 33%, Adjusted EBITDA1 of $24.7 million, Adjusted EBITDA margin of 19% and net income before tax was $6.8 million
- Year-end Cash and Equivalents of $38.6 million
FOURTH QUARTER 2019 SUMMARY1
- Revenue increased to $32.4 million from $10.2 million in Q4 2018, and decreased 25% from $43.4 million in Q3 2019 due to a reduction in volume of bulk extract sold and average selling price
- Gross profit increased to $10.0 million from $4.0 million in Q4 2018 and decreased 32% from $14.7 million in Q3 2019. Gross margin was 31% compared to 39% a year ago and down from 34% in Q3 2019 primarily due to a reduction in the average selling prices and impact of lower volumes on production costs, partially offset by lower dried flower costs
- Adjusted EBITDA(1) increased to $2.7 million in Q4 2019 from $2.1 million in Q4 2018, and decreased 74% from $10.1 million in Q3 2019. The decrease from Q3 2019 reflects a decrease in revenue and gross profit and increased operating expenses attributable largely to ERP implementation expenses
- Net loss before tax decreased to $2.4 million from $3.5 million in Q4 2018, and decreased from net income before tax of $5.4 million in Q3 2019
- As at December 31, 2019, past due balance of receivables was approximately $8.6 million, of which $7.7 million was related to a previously announced statement of claim filed subsequent to quarter end. All remaining outstanding balances as at year-end have been collected
The Company reported that the Canadian cannabis sector experienced significant challenges in the fourth quarter that resulted in an oversupply of bulk extract in the market and subsequent reduction in selling price and lower volumes. These factors included: a slower than anticipated expansion of retail channels in Canada, slower licensing of new and specialized cannabis 2.0 businesses, lower than expected conversion of bulk extract inventory into finished goods by vertically integrated players and slower than expected entry to the market by larger consumer packaged goods and pharmaceutical companies.
“Through 2019, MediPharm Labs pioneered its way in an incredibly young industry. We executed well on a highly specialized and focused business model and took a leadership position among the top five cannabis revenue generating companies in Canada,” said Mr. McCutcheon. “We ended the year with capital strength and liquidity, while achieving and surpassing all of our significant operational and financial milestones despite the significant challenges faced by the sector in Canada.”
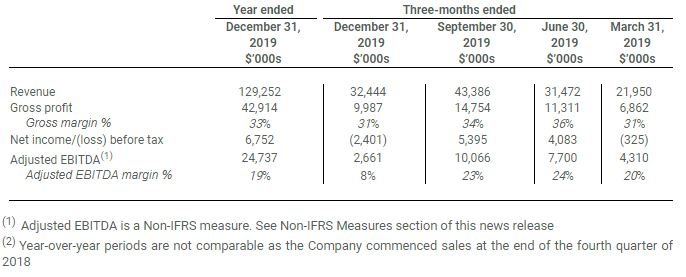 (1) Adjusted EBITDA is a Non-IFRS measure. See Non-IFRS Measures section of this news release
(1) Adjusted EBITDA is a Non-IFRS measure. See Non-IFRS Measures section of this news release
(2) Year-over-year periods are not comparable as the Company commenced sales at the end of the fourth quarter of 2018
Looking Ahead
The cannabis market challenges experienced in the fourth quarter are expected to persist in the near-term. Additionally, while it remains a rapidly evolving situation, the global COVID-19 pandemic is expected to have various operational impacts in the near term, including potential supply chain challenges, work from home adjustments, and enhanced safety protocols. The Company’s manufacturing facility was exempted from the recent provincial mandated workplace closures as an essential service, the extent of the impact on COVID-19 on the Company’s operational and financial performance will depend on various developments, including the duration and magnitude of the outbreak, and the impact on customers, employees and vendors.
.As a result, the Company has taken active steps in the immediate term to manage the business and maintain its liquidity and financial strength enabling management to continue prudently develop and invest in the business for the longer-term. In the immediate term, the Company is executing on the following:
Diversifying and Accelerating Growth through:
- Prioritizing and accelerating new product development opportunities already in the Company’s pipeline of 2.0 products and outside the current portfolio – including topicals or R&D for other delivery methods
- Launch of in-house MediPharm branded wellness and medical products further diversifying the Company’s revenue stream and targeting distribution across multiple patient and adult-use channels
- Intensified efforts to supply international medical markets and enhance global distribution, now possible given receipt of first GMP
- Entering new contract manufacturing agreements to enhance customer’s ability to convert bulk extract inventory into finished goods
Maintaining Liquidity and Financial Strength through:
- Deferral of non-essential budgeted capital expenditures
- Reduced employee base by 10% in Q1 and rebalanced production workforce to meet needs of current environment and future growth
- Procurement driven cost savings initiatives whilst continuing to focus on improving operational efficiencies
“As an expression of confidence in the longer-term potential of our Canadian footprint, we are moving forward with our recently launched in-house brand and to scale up our automated downstream production, quality control and testing capabilities, as well as our research and development functions within our recently expanded GMP facility in Barrie,” said Mr. McCutcheon. “These initiatives will support our brand as well as the fulfillment and distribution of orders and new product formulations not only in 2020 but in the decade ahead.”
“With our first GMP certification in hand, we are well positioned to supply international medical markets that offer additional growth and diversification for our revenue streams as the Canadian market continues to evolve,” said Mr. McCutcheon. “Our international efforts are expected to be further strengthened by efforts to broaden our GMP certifications to include Europe and the start-up of our Australian facility.”
Mr. McCutcheon added: “While current domestic market conditions are disappointing, MediPharm Labs has attracted a number of exciting customers and prospects both internationally and domestically, proving that our expertise and GMP-certified capabilities are in demand.”
GROWTH CATALYSTS
- Expected EU GMP certification of the Canadian facility and TGA GMP certification of Australian facility
- International export agreements
- Development of high-quality branded in-house consumer products and contract manufacturing agreements
- Application of MediPharm Labs’ Research License
- New opportunities for R&D and clinical trials
The Company’s consolidated financial statements and management’s discussion and analysis for the three and nine months ended December 31, 2019 are available on SEDAR and on www.medipharmlabs.com.
Q4 CONFERENCE CALL AND WEBCAST
The Company will host a conference call and audio webcast on March 30, 2020 at 8:30 a.m. eastern time to discuss its results and outlook. Participants are asked to dial in approximately 10 minutes before the start of the call using one of the following numbers: Toll-free: 877-791-0216; International: 647-689-5661.
A replay will be available approximately one hour after the call ends by dialing: Toll-free: 800-585-8367; International 416-621-4642. Conference ID: 5576297.
An audio webcast will be available in the Events section of the MediPharm Labs’ Investor Relations website https://ir.medipharmlabs.com/news-events or by visiting the following link: https://event.on24.com/wcc/r/2152211/3D5EEC47DD1A08B3183F90AD781074FE
About MediPharm Labs Corp.
Founded in 2015, MediPharm Labs specializes in the production of purified, pharmaceutical-grade cannabis oil and concentrates and advanced derivative products utilizing cGMP (current Good Manufacturing Practices) designed facility and ISO standard built clean rooms. MediPharm Labs has invested in an expert, research-driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with 5 primary extraction lines having 300,000 KG of annual processing capacity to deliver pure, safe and precisely-dosed cannabis products for its customers. Through the Company’s wholesale, private and white label platform, MediPharm Labs formulates, processes, packages and distributes active pharmaceutical ingredients and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm Labs has completed commercial exports to Australia and is nearing completion of its Australian extraction facility expected in 2019 with 75,000 KG of annual processing capacity.
For more information:
Laura Lepore, VP, Investor Relations
Telephone: +1 416.913.7425 ext. 1525
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
