MediPharm Labs Reports First Quarter 2020 Results, Clears Path for International Growth
BARRIE, Ontario, June 18, 2020 (GLOBE NEWSWIRE) — MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven cannabis extraction, distillation and derivative products, today announced its financial results for the three months ended March 31, 2020. The Company also provided an update on the build out of its global Good Manufacturing Practices (“GMP”) certified production platforms and new international medicinal, wellness and adult use-recreational revenue streams.
“MediPharm Labs has proudly evolved from our beginning stages to today, capitalizing on a multi-jurisdictional GMP-certified manufacturing footprint to further develop a diversified international customer portfolio with multiple product streams suitable for lawful medicinal, wellness and adult-use recreational markets around the world,” said Pat McCutcheon, CEO, MediPharm Labs. “Our focus on innovation, a pharmaceutical approach, capital investments and the very high standards achieved through the GMP certification of our Canadian and Australian facilities will allow us to further evolve our business as we pursue medicinal and wellness markets, as well as international opportunities, so that we are not reliant on the challenged Canadian recreational market.”
Due to a reduction in the average selling price of bulk extracts and reduced volumes sold because of a continued muted demand in the Canadian bulk wholesale market, first quarter revenue was 49% below the same quarter a year ago.
We successfully scaled our B2B bulk business in the lead up to Cannabis 2.0 legalization where demand for bulk cannabis concentrates soared; but the limitations of a single regulated market, with limited distribution and slow finished product roll-out, resulted in a decrease in demand for those bulk concentrates beginning in Q4 2019 and continuing to today.
 Pat McCutcheon, CEO, MediPharm Labs
Pat McCutcheon, CEO, MediPharm Labs
This is why our vision of operating a global platform for multi-customer, multi-product and multi-jurisdictional relationships covering medicinal, wellness and adult-use segments makes sense from strategic, risk management and value creation perspective as the Canadian market for concentrate-based products continues to mature.
“Our capital investments in our two GMP-platforms have been made creating further differentiation from our competitors around the world and we are developing relationships in various countries for a range of cannabis products, ” said McCutcheon.
BUSINESS HIGHLIGHTS AND ACCOMPLISHMENTS
- Near-Term Australian Commercialization: During the first quarter, MediPharm Labs Australia secured a white label supply agreement with Compass Clinics Australia Pty Ltd. and subsequent to quarter end, secured white label supply agreements with, among several others, Burleigh Heads Cannabis Pty Ltd. (Australia); Helius Therapeutics Limited (New Zealand); Cannasouth Plant Research New Zealand Limited (New Zealand); and Therismos Limited (UK). Sales of GMP-certified formulated products are expected to begin within the second quarter of 2020 and ramp up in Q3 2020 and beyond.
- GMP Certification: Subsequent to quarter end, the Company’s Australian facility was certified by the Therapeutic Goods Administration (“TGA”) as meeting the GMP standard and secured a Licence to Manufacture Therapeutic Goods. As the Company’s Canadian facility was TGA GMP certified in late 2019, MediPharm Labs has created a global pharmaceutical-quality supply chain that is qualified to serve new emerging medical markets internationally.
- Increasing Diversification of Product Mix: During the first quarter, the Company launched its house brand with the introduction of high-quality CBD oils and shipped its first topicals to a contract manufacturing customer. Subsequent to quarter end, it entered into a strategic manufacturing and intellectual property licensing agreement with Biopharmaceutical leader, Avicanna Inc. through which it intends to commercialize a diverse array of sophisticated product formats.
- Canadian Medical Channel Sales: The Company announced that it was selected by Shoppers Drug Mart to supply high-quality concentrate products to medical patients through the national Medical Cannabis by Shoppers™ online platform. The ability to distribute through this channel gives the Company direct exposure to Canada’s medical market, without having to establish its own medical sales infrastructure.
- Finished Formulated Product Sales: By the end of the first quarter of 2020, the Company had successfully launched three new SKUs of consumer packaged cannabis-based goods across five provinces. Sales of finished formulated products grew to comprise 13% of first quarter revenue, up from nil in the fourth quarter of 2019. The proportion of sales comprised of finished formulated products is expected to increase due to an expansion of the Company’s domestic and international sales team and the launch of eight additional SKUs subsequent to quarter end. The continued expansion of finished formulated products manufacturing and distribution capabilities is expected to increase sell-through of bulk concentrate inventory. To date in Q2 2020, the Company has already shipped more than double the quantity of finished formulated products that it shipped in all of Q1 2020.
- Bulk Crude Resin and Distillate Sales and COVID-19: Canadian B2B market sales continued to decline in the first quarter of 2020 as a result of oversupply in the Canadian bulk crude resin and distillate markets, given a slow expansion of retail channels in Canada, slow commercialization of new and specialized Cannabis 2.0 businesses and operational challenges from B2B customers seeking to convert bulk concentrates into consumer packaged goods. The significant challenges experienced in the Canadian B2B market were further exacerbated by the impact of the COVID-19 pandemic, including additional delays in the anticipated expansion of retail channels in Canada, slower licensing of new cannabis derivative businesses, and delays in closing new contracted customers.
Q1 FINANCIAL SUMMARY
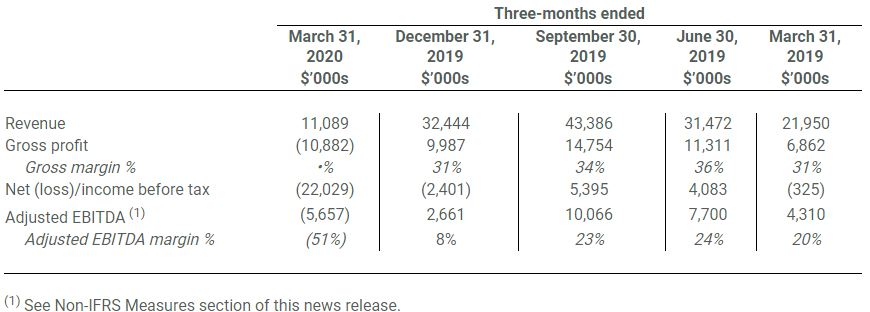
- Revenue was $11.1 million in Q1 2020, a 49.5% decrease over Q1 2019 due to a reduction in volume and average selling price of bulk concentrates sold, which was partially offset by growing sales of consumer packaged goods to provincial distributors throughout Canada. Going forward, the Company expects its inventory procurement costs to shrink reducing overall cost of goods.
- Gross profit was ($10.9 million) compared to gross profit of $6.9 million in Q1 2019 primarily due to a $12.8 million write down of inventory to net realizable value, owing to the continued oversupply in the Canadian domestic bulk concentrates market, and a reduction in the average selling price and volume of bulk concentrates sold, partially offset by lower dried flower input costs. Gross profit before the inventory write down was $1.9 million (gross margin of 17%).
- Net loss before tax of $22 million in Q1 2020 was largely attributable to the $12.8 million write down of inventory to net realizable value, reduced average selling prices and a share-based compensation expense of $2.8 million.
- Adjusted EBITDA (1) was ($5.7 million) in Q1 2020 compared to $4.3 million in Q1 2019 due an increase in headcount and ERP implementation expenses relative to the Company’s early stage of commercialization in Q1 2019.
- Cash and equivalents balance at the end of March 31, 2020 was $21.4 million. Subsequent to quarter end, the Company successfully completed a $37.8 million private placement, with half of such gross proceeds remaining in escrow as of today.
REDUCING COSTS TO ADDRESS COVID-19
“COVID-19 exacerbated the challenge of establishing a strong legal cannabis market in Canada by delaying retail store openings, making it difficult for vertically integrated producers to ramp up capabilities to convert bulk extract into finished products, and making new customers challenged to commit to longer term agreements in an economically uncertain environment,” said Bobby Kwon, Chief Financial Officer. “This reduced product ordering and sell through has negatively impacted our Q1 revenue and capacity utilization. In response, we have made strong progress on ramping up consumer packaged goods production to diversify our revenue streams while actively reducing and managing expenses to maintain a strong cash balance.”
“The Canadian market is still developing and MediPharm Labs will be the beneficiary as retail channels and available products expand. But we are not waiting, which is why we have consistently prioritized investments in, and expansion of, our international medicinal and wellness markets.”
MediPharm Labs is an essential business and as such, its Canadian manufacturing facility was exempted from provincially mandated COVID-19 closures. While the Company continues to maintain adequate inventory to supply its customers and meet demand, vertically integrated organizations have been slower to add capabilities to convert bulk extract into finished product resulting in lower-than-historical demand for bulk wholesale extracts. As a result of the pandemic, these customers may now also be experiencing further business restrictions and interruptions as well as disruptions through retail distributors which may lead to reduced product ordering and sell through.
STRENGTHENING LIQUIDITY
As announced on June 8, 2020, the Company completed a private placement, raising aggregate gross proceeds of $37.8 million, 50% of which are being held in escrow pending satisfaction of certain conditions. This substantially improved the Company’s liquidity and ability to fully support its strategies including exporting products to new international jurisdictions and launching consumer packaged cannabis-based offerings with new formulations and formats.
LOOKING AHEAD
The Company expects Canadian market inefficiencies – including those related to COVID-19 – to persist but slowly moderate while international sales agreements begin to ramp up. Accordingly, it anticipates that second half 2020 results will be stronger than first half 2020 performance. To improve performance, the Company will execute on the following priorities:
Diversifying and igniting growth by:
- Developing quality earnings from a wide spectrum of international cannabis markets including medicinal, recreational and wellness.
- Delivering high-quality consumer packaged products under white label agreements with leading LPs, biopharmaceutical and consumer products companies.
- Entering into the clinical trials arena to broaden medicinal and wellness applications.
- Broadening its in-house branded product portfolio and capturing consumer market share with the newly launched MediPharm Labs family of products.
Maintaining liquidity and financial strength by:
- Capitalizing on the value of the Company’s international supply chain (including procurement sources) and build out of its multi-jurisdictional manufacturing capability to realize cost and production efficiencies.
- Closely managing all expenses through a continuation of cost containment measures implemented in Q1 and Q2 to date.
- Disciplined capital deployment and continuing to defer non-essential, historically budgeted capital expenditures.
“In light of the current market environment, we remain confident in our core strategies and business model. We believe our approach remains entirely appropriate for the creation of long-term customer and shareholder value,” said Mr. McCutcheon. “With the right framework in place, all the hard work already done by our team to invest in and build two high-end, pharmaceutical-grade facilities that are GMP certified, and a culture devoted to continuous improvement, I am convinced that MediPharm Labs has everything we need to succeed as a diversified global provider of differentiated medical, wellness and adult recreational use cannabis products.”
GROWTH CATALYSTS
- Revenue from recently secured international white label medicinal and wellness supply agreements.
- Commercialization of Australian extraction facility and formation of additional customer supply agreements.
- Clinical trials and expansion of addressable medicinal and wellness markets through new indications.
- Further development of MediPharm Labs branded high-quality consumer products.
- Continued rollout of the Canadian retail market.
The Company’s consolidated financial statements and management’s discussion and analysis for the three months ended March 31, 2020 will be available on SEDAR and on www.medipharmlabs.com
Q1 CONFERENCE CALL AND WEBCAST
The Company will host a conference call and audio webcast on Thursday, June 18, 2020 at 8:30 a.m. Eastern Time to discuss its results and outlook.
Conference Call Information:
Toll-free number: 833-502-0471 / International number: 236-714-2179 / Conference ID: 5073099
Due to higher than normal volumes, participants are asked to dial in approximately 15 minutes before the start of the call.
Audio Webcast:
An audio webcast will be available in the Events section of the MediPharm Labs’ Investor Relations website https://ir.medipharmlabs.com/news-events or by visiting the following link: https://event.on24.com/wcc/r/2159388/A36D3A773AE4FDB8F068C0A4C5024CB0
For those who are unable to participate on the live conference call and webcast, a replay will be available approximately one hour after completion of the call.
Replay Information:
Replay number: Toll-free number: 800-585-8367 / International replay number: 416-621-4642/ Conference ID: 5073099
NON-IFRS MEASURES
Adjusted EBITDA is not a recognized performance measure under IFRS, does not have a standardized meaning and therefore may not be comparable to similar measures presented by other issuers. Adjusted EBITDA is included as a supplemental disclosure because Management believes that such measurement provides a better assessment of the Company’s operations on a continuing basis by eliminating certain non-cash charges and charges or gains that are nonrecurring. Adjusted EBITDA is defined as net loss excluding interest, taxes, depreciation and amortization, share-based compensation, and other non-cash expenses. Adjusted EBITDA has limitations as an analytical tool as it does not include depreciation and amortization expense, interest income and expense, taxes, share-based compensation and transaction fees. Because of these limitations, Adjusted EBITDA should not be considered as the sole measure of the Company’s performance and should not be considered in isolation from, or as a substitute for, analysis of the Company’s results as reported under IFRS. The most directly comparable measure to Adjusted EBITDA calculated in accordance with IFRS is operating income (loss). The above is a reconciliation of the Company’s operating loss to Adjusted EBITDA. See “Reconciliation of non-IFRS measures” in the Company’s Management’s Discussion and Analysis for the period ended March 31, 2020 for additional information.
About MediPharm Labs
Founded in 2015, MediPharm Labs specializes in the production of purified, pharmaceutical-quality cannabis oil and concentrates and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm Labs has invested in an expert, research- driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with five primary extraction lines for delivery of pure, trusted and precisely-dosable cannabis products for its customers. Through its wholesale and white label platforms, MediPharm Labs formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm Labs has completed commercial exports to Australia and is nearing commercialization of its Australian extraction facility. MediPharm Labs Australia was established in 2017.