
VIVO Cannabis™ Reports Q2 2019 Financial and Operating Results
- Net revenue was $5.3 million in the quarter, a five-fold increase over the same quarter last year.
- Cultivation license was granted in the third quarter of 2019 for four new seasonal greenhouses in Napanee Ontario, increasing VIVO’s annualized cultivation capacity by 4,000 kilograms and contributing to its innovative low-cost capacity plan to supply a broad line of products, including cannabis infused edibles, topicals and beverages.
- Current licensed internal cultivation capacity now totals 12,000 kilograms and agreements are in place with two licensed third-party cultivators to supply approximately 5,000 kilograms per annum.
- Health Canada approval was received to begin extraction operations using VIVO’s on-site CO2 extraction system at the Napanee facility, and the Company has procured an ethanol system that will bring total extraction capacity to 50,000 kilograms per year.
- Significant progress was made on all four of VIVO’s strategic priorities with capital being allocated to production, product development and international expansion – all important initiatives that facilitate future profitable growth.
NAPANEE, ON, Aug. 28, 2019 /CNW/ – VIVO Cannabis Inc. (TSX-V: VIVO, OTCQX: VVCIF) (“VIVO” or the “Company”), today released its second quarter 2019 financial and operating results.
The progress our team has made on all four of our strategic priorities positions us well for future growth. We remain focused on increasing long-term shareholder value by continuing to establish ourselves as a premium provider of cannabis products and services. Our positive momentum is expected to lead to increased revenue generation and product breadth in the second half of the year.
 Barry Fishman, Chief Executive Officer of VIVO
Barry Fishman, Chief Executive Officer of VIVO
VIVO’s net revenue increased slightly to $5.3 million during the second quarter of 2019 compared to the first quarter of 2019 as product supply remained essentially flat. The harvesting and packaging of the Company’s increase licensed capacity is expected to result in growth in net revenue in the second half of 2019. The Company also continued to sustain top-tier prices with a net average selling price of $6.96 per kilogram of dry flower in the second quarter.
Adjusted EBITDA was ($1.2) million in the second quarter compared to ($2.2) million in the first quarter. As at June 30, 2019, cash and short-term investments (excluding strategic equity investments) were approximately $59 million.
Key Financial Results
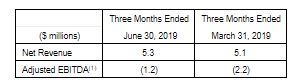
¹Adjusted EBITDA is not a measure of financial performance under IFRS. The definition of Adjusted EBITDA, which excludes unrealized gains and losses related to strategic investments, can be found in the Company’s management’s discussion and analysis for the three and six months ended June 30, 2019, available under the Company’s profile at www.sedar.com
Additional details with respect to VIVO’s financial results are available in the Company’s management’s discussion and analysis and interim financial statements for the period ended June 30, 2019, both of which can be found on SEDAR at www.sedar.com.
Strategic Priorities
VIVO made significant progress on all four of its strategic priorities during the second quarter of 2019. The Company believes that the attainment of its strategic goals will help grow long-term shareholder value by establishing VIVO as a premium provider of cannabis products and services that enhance people’s lives.
The strategic priorities are as follows:
1. Enhance supply and production capabilities
In the first quarter of 2019, VIVO doubled its annual cultivation capacity at both the Canna Farms facility and the Vanluven facility from a total of 4,000 kilograms to 8,000 kilograms. VIVO currently has agreements to purchase approximately 5,000 kilograms per annum of domestic and international third-party supply for the Canadian and international markets.
Subsequent to the end of the second quarter of 2019, VIVO was granted a standard cultivation licence from Health Canada for its Kimmetts facility in Napanee, Ontario – a hybrid outdoor cultivation site consisting of four seasonal greenhouses and an associated header house that increases the Company’s annualized internal cultivation capacity to 12,000 kilograms. Also, in the third quarter of 2019, VIVO received approval from Health Canada to commence extraction operations at its Vanluven facility using its in-house supercritical CO2 extraction suite. The Company recently began the installation of a large-scale ethanol extraction system at its Vanluven facility and expects new distillation systems to be installed in the third quarter of 2019. Once these systems are fully operational, the Vanluven facility is expected to be capable of processing more than 50,000 kilograms of cannabis per year. VIVO has also invested in automated packaging and labelling equipment, expected to significantly increase finished good throughput and lower manufacturing costs.
The Company has also commenced construction of Phase 5 of its Canna Farms facility. This expansion is expected to cost approximately $3.5 million and bring online an additional 2,500 kilograms of capacity when completed in early 2020, bringing total internal and third-party cultivation capacity to approximately 20,000 kilograms.
2. Create a broad and loyal distribution network
VIVO will continue to serve both the medical and adult-use markets through its collection of premium, trusted brands, including Beacon Medical™, Fireside™, Canna Farms™, Lumina™ and Harvest Medicine. In April 2019, Canna Farms received the strong support of its patients, winning the People’s Choice Award for “Best LP Customer Service” at the O’Cannabiz Industry Gala.
VIVO has continued to build upon the Canna Farms medical cannabis platform launched in the first quarter of 2019 (https://www.cannafarms.ca) where patients can access one of the widest selections of cannabis products in the Canadian medical market. Additionally, licensed producers can sell their quality products through this marketplace and make a fair return while avoiding the complex processes necessary for medical cannabis fulfillment to patients. The platform, which has increased patient registrations by over 10% in the past three months, now has 19,000 registered patients and provides access to products cultivated by four different licensed producers.
Through its brick and mortar clinics and HMED Connect telemedicine application, Harvest Medicine has conducted over 50,000 patient visits, making it one of the top clinic networks in Canada. Harvest Medicine operates five medical cannabis clinics in the provinces of Alberta, New Brunswick, Nova Scotia and Ontario, and continues to evaluate new clinic locations. Harvest Medicine’s business continues to expand, focusing on providing cannabis-focused education to patients along with other complementary health services and support.
In the adult use market, VIVO has entered into strategic partnership agreements with leading Canadian cannabis retailers, National Access Cannabis Corp., Choom Holdings Inc., and Westleaf Cannabis Inc. In the second quarter of 2019, VIVO entered into a strategic partnership with Friendly Stranger Holdings Corp., a well-known player in the cannabis accessory and lifestyle market. VIVO currently has adult-use supply agreements with the provinces of British Columbia, Alberta, Saskatchewan, Manitoba, Ontario and the Yukon, and anticipates having secured agreements with most or all provinces by the end of 2019.
3. Build an innovation-driven culture and portfolio
Subsequent to the end of the second quarter of 2019, VIVO was granted a cultivation licence from Health Canada for its Kimmetts facility in Napanee, Ontario. VIVO is the only Canadian licensed producer employing this innovative airhouse technology which is currently used in several US states to produce high-quality cannabis at low cost without the capital investment required to build traditional greenhouses or indoor grows. The remaining 55 acres at the Kimmetts property have the potential to accommodate significant additional cultivation capacity.
Also subsequent to quarter-end, VIVO’s Vanluven facility received approval from Health Canada to commence extraction operations using its supercritical CO2 extraction suite. This suite, in conjunction with the ethanol extraction and distillation processes the Company is implementing, will be used to produce high quality oils, distillates, concentrates and more advanced formulations for VIVO’s current and anticipated portfolio of medical products, as well as edibles, beverages and topicals for the recreational market in late 2019 at a fraction of the cost of using third-party contract extraction services.
VIVO’s GMP-designed Vanluven facility is conducting ongoing research related to the development of optimized indoor, seasonal greenhouse and outdoor grown cannabis cultivars in conjunction with the University of Guelph. The “Guelph R&D room” in the Vanluven facility is on the cutting-edge of technology using six PS1000 growth chambers where experiments are ongoing to improve cultivation yields and growing techniques.
The Vanluven facility, through its multi-year collaboration with Loyalist College, is evolving into a centre of excellence for the extraction and distillation of cannabis oils, and formulation expertise in the development of cannabis infused edibles, topicals and beverages.
The Company has definitive agreements with four Canadian and U.S. based product development partners with expertise in the respective field of endeavor towards the goal of creating novel formulations and delivery formats. Of these partners, the Company has only currently disclosed its collaboration with Pharmascience, a global pharmaceutical company based in Montreal, Quebec. Overall, the Company’s goal is to offer medical patients and health care professionals a broad range of innovative, high-quality forms of cannabis.
VIVO, through its Australian subsidiary and a leading pain clinic in Melbourne, Australia, is conducting a study looking at the use of cannabis for treating chronic pain.
4. Accelerate international market growth
VIVO continues to focus on targeted international expansion activities and intends to focus on select markets in which medical cannabis can be legally prescribed, leveraging the strong pharmaceutical background of its leadership team, its experience operating Harvest Medicine clinics, and its work with patients who have benefited from medical cannabis, to increase its footprint and become a leader in the global medical cannabis sector.
The Company entered into a definitive supply agreement with a European cultivation partner in early 2019 and currently has bulk product produced from VIVO’s genetics that will be available for sale in Europe, pending EU-GMP and regulatory approval. Using the supercritical CO2 extraction technology in Napanee, the Company expects to be able to ship extracted cannabis products internationally for medical purposes, subject to the receipt of EU-GMP certification and other approvals.
VIVO’s German subsidiary, ABcann Germany GmbH, has successfully undergone an inspection by regional health authorities and was awarded authorization to import medical cannabis from non-EU countries into Germany. This authorization comes on top of ABcann Germany’s wholesale pharmaceutical license, which was received in the first quarter of 2019. ABcann Germany has filed an application to receive a narcotics license from the Federal Institute for Drugs and Medical Devices. When its suppliers receive EU-GMP certification, ABcann Germany will be fully approved to commercialize and sell its medical cannabis products.
VIVO expects its international business to begin operating under the “Beacon Medical™” brand in late 2019. The Company is exploring additional opportunities in select European markets where medical cannabis has been legalized, including in the UK and Poland.
VIVO’s Australian business continues to grow. The Company has shipped product to Australia from both its Canna Farms and Vanluven facilities in 2019. In the second quarter of 2019, VIVO entered into a distribution agreement with Burleigh Heads Cannabis Pty Ltd, one of Australia’s leading medicinal cannabis distributors. The agreement is expected to increase the ability of Australian patients to access VIVO’s high-quality medicinal cannabis flower, oil and other products via approved channels.
Near-term Catalysts
VIVO has identified the following positive investment catalysts that it is on track to achieve in the near- term:
- EU-GMP certification – VIVO’s Vanluven facility is nearing EU-GMP certification, which is the global gold standard in the pharmaceutical industry and speaks to VIVO’s commitment to a quality-first culture for its facilities and products. A quality and compliance focus is part of VIVO’s DNA.
- Domestic product line expansion –The expansion of Canada’s cannabis market to include edibles, beverages, concentrates and topicals, targeted for December 2019, represents a significant opportunity for all market participants. VIVO has entered into a partnership with a former Belgian chocolatier who is currently operating an award-winning company called ChocolaTas to produce premium cannabis chocolates at the Canna Farms facility in Hope B.C. The Company expects to have product available for sale as soon as practicable following the opening of the edibles market. The Company has new product lines that it intends to make available for this market and has submitted a targeted but significant portfolio of ‘Phase II’ innovative products to various provinces in June in response to product calls.
- Increased extraction capability – VIVO now has 12,000 kilograms of internal dry flower cultivation capacity and annualized commitments of approximately 5,000 kilograms from third parties. The Company expects to increase its extraction capacity to approximately 50,000 kilograms of dry flower input during 2020.
- Seasonal greenhouse harvest – After the granting of a standard cultivation licence from Health Canada for its Kimmetts facility and completion of current planting, VIVO expects to realize its firstharvest from its seasonal greenhouses in the fourth quarter of 2019.
- Approval to sell in Germany – VIVO received its pharmaceutical wholesale licence and EU-GMP-certificate from the regulatory authorities of the state of Brandenburg, Germany. The Company expects its narcotic and import license approvals in early 2020, which will facilitate imports of higher-margin products to the European medical market from other countries, including Canada.
Adoption of Minimum Share Ownership Policy and Automatic Share Disposition Plan
The Company is pleased to announce that it is adopting a Minimum Share Ownership Policy (the “Policy”). The goal of the Policy is to align the economic interests of the Company’s senior officers and directors with those of the Company’s shareholders. Under the terms of the Policy, directors and senior officers will be required to hold shares having a value equal to a multiple of their annual cash retainers. The required ownership levels are required to be attained by each participant within five years of becoming subject to the Policy.
In addition, Daniel Laflamme, a director and senior officer of the Company, and one of its largest shareholders, has entered into an automatic securities disposition plan (the “Plan”), filed on SEDAR. The Company understands that a sizeable amount of Mr. Laflamme’s personal net worth is represented by his shares in the Company and appreciates his desire to liquidate a small portion of his equity position while minimizing any potential negative impact in the Company’s share price.
“Through my role as a director, Chief Operations Officer and significant shareholder, and a pioneer of the legal cannabis industry, I am extremely committed to, and involved in, every aspect of VIVO’s business and success,” said Mr. Laflamme. “After seeing how large investors can affect a company’s share price, I believe the Plan will allow me to liquidate a small portion of my investment to support my personal life, while still allowing me to focus on building VIVO’s business and avoiding large dispositions in the brief windows where I would otherwise be permitted to trade.”
Canadian securities laws and the Company’s Insider Trading Policy place significant restrictions on when insiders of the Company can buy and sell shares. The Plan will permit Mr. Laflamme to have shares sold on his behalf by an independent securities broker on an automatic basis, regardless of any material undisclosed information he might receive in the future.
Under the Plan, up to 100,000 shares per week (up to 5.2 million total over the 12-month term of the Plan), can be sold, representing less than 2% of the Company’s currently issued and outstanding shares. Sales will only be executed if the amount to be sold represents less than 30% of the previous day trading volume.
Mr. Laflamme will not exercise any discretion or influence over how sales of the Shares governed by the disposition plan will occur. Mr. Laflamme will still be permitted to purchase or dispose of Shares outside of the Plan, subject to compliance with the Company’s Insider Trading Policy and applicable securities laws. The Plan will become effective on September 1, 2019.
The Company intends to apply to applicable securities regulators for an exemption from insider trading disclosure requirements with respect to sales under the Plan. Unless and until such an exemption is obtained, transactions under the Plan will be reported on SEDI at www.sedi.ca in accordance with applicable Canadian securities laws. Each such filing will bear a notation to advise readers that the dispositions relate to the Plan.
Q2 2019 Results Conference Call and Webcast
DATE:
Thursday, August 29, 2019
TIME:
10:00 am ET
DIAL-IN NUMBER:
647-427-7450 or 1-888-231-8191
CONFERENCE ID:
4479716
LIVE WEBCAST:
About VIVO Cannabis™
VIVO, based in Napanee, Ontario, is recognized for trusted, premium cannabis products and services. It holds production and sales licenses from Health Canada and operates world-class indoor cultivation facilities with proprietary plant-growing technology at its Canna Farms facility in Hope, B.C., and at its Vanluven facility in Napanee, Ontario. VIVO has a collection of premium brands targeting unique customer segments, including Beacon Medical™, Fireside™, Canna Farms™ and Lumina™. The Company is significantly expanding its production capacity and distribution channels; growing its domestic medical cannabis platform, including Harvest Medicine, its patient-centric, highly scalable network of specialty medical cannabis clinics and services; promoting production and cultivation innovation and pursuing partnership and product development opportunities; and actively focusing on growth in select international markets, including Germany and Australia. VIVO has a healthy balance sheet and is well-positioned to accelerate its growth in Canada and internationally. For more information visit: www.vivocannabis.com
