Last week, GW Pharma (NASDAQ: GWPH) scored a milestone victory, as the thirteen-member panel at the Peripheral and Central Nervous System Drugs Advisory Committee, which met on April 19th to consider the safety and efficacy of Epidiolex, its CBD-based product, and pass along its conclusions to the FDA, gave it a unanimous decision in favor of Epidiolex. The committee, which referred to Epidiolex as “Cannibidiol Oral Solution” (CBD-OS), saw it as safe and efficacious in treating seizures in patients 2 years of age and older suffering from Lennox-Gastaut syndrome and Dravet syndrome. The FDA is expected to complete its review process of the Epidiolex New Drug Application by June 27th. If approved, Epidiolex will be the first cannabis-derived FDA-drug approved.
Clearing a Big Misconception
Many are curious about how FDA approval of a cannabis-derived medicine will impact scheduling of cannabis, which is a Schedule 1 controlled substance having “no currently accepted medical use and a high potential for abuse,” according to the Drug Enforcement Agency (DEA). While the approval of Epidiolex will certainly lay the foundation for a challenge to the DEA scheduling of the underlying plant from which CBD is derived or even CBD itself, the DEA can schedule it differently from cannabis itself. GW Pharma expects the drug, if approved, to be Schedule 4, similar to many sleep or pain medications. The DEA will have up to 90 days upon FDA approval to schedule Epidiolex, assuming Health and Human Services provides it with a recommendation concurrently with the FDA approval.
The law further states that the 90-day time-frame starts the later of (1) the date DEA receives the HHS scientific and medical evaluation/scheduling recommendation or (2) the date DEA receives notice of the NDA approval by HHS. In addition, the law specifies that the rule-making shall become immediately effective as an interim final rule without requiring the DEA to demonstrate good cause therefor.
Most recently, the DEA took more than 90 days to schedule Syndros, a synthetic THC oral solution developed by Insys Therapeutics, that was approved by the FDA on July 1, 2016. The DEA issued an interim ruling on March 23, 2017 that was finalized in late 2017, though this delay was likely because of the abuse potential of Syndros, a rapid-acting product, that required additional information.
CBD Products are Booming
Brightfield Group, a market research firm based in Chicago, expects CBD products to grow from 5% of U.S. cannabis sales today to 12% by 2021, and this doesn’t include the pharmaceutical channel that would open up with the approval of Epidiolex. The firm tracks several different distribution channels and suggests that total CBD sales of $288 million in 2017 grew 65% and predicts the entire category to grow 76% in 2018 to $505 million. In 2017, it estimates the share of each distribution channel:
- Dispensaries and delivery services: 7%
- Health shops: 6%
- Smoke shops: 21%
- Doctor’s offices: <1%
- Online sales: 65%
Will the Approval of Epidiolex Doom Sale of CBD Products?
Most of the demand for CBD is from consumers or patients who suffer from anxiety, insomnia or sleep problems, joint pain and inflammation and depression, according to a study conducted by Brightfield Group and HelloMD. In their survey, they learned that only 2% of the respondents cited epilepsy as the medical condition they were seeking to relieve:
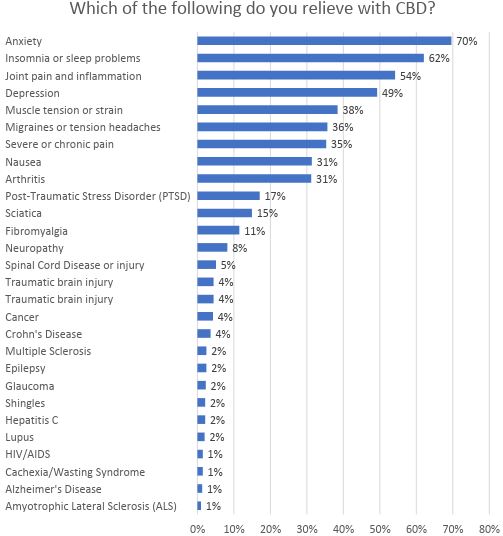
With only a small amount of the overall CBD business likely tied to the treatment of epilepsy, Epidiolex doesn’t appear to offer a threat to current CBD providers, except perhaps in that narrow category. Epidiolex, if approved, will be indicated only for patients suffering from LGS or Dravet syndrome, though doctors would be able to prescribe it for additional indications. Most analysts expect that there could be significant off-label activity, as doctors treat patients who suffer from other forms of epilepsy or possibly other indications. GW Pharma is studying Epidiolex to treat tuberous sclerosis complex and infantile spasms.
One of the big advantages of Epidiolex is that it will likely be covered by insurance companies, a distinct advantage over traditional medical cannabis that will likely overcome any sort of cost advantage that dispensary or mail-order CBD might offer. Epidiolex is expected to have an annual cost of $30K or more. Additionally, quality control is likely to push potential patients to adopt FDA-approved Epidiolex over other sources of CBD.
A great example of how the market may evolve with both prescription CBD and over-the-counter CBD is the fish oil market. Lovaza is a prescription-based drug marketed by GlaxoSmithKline and now available generically that was first approved by the FDA in 2004, and there are now several others, including Vascepa, Epanova and Omtryg. The OTC market for fish oil remains vibrant despite the creation of a pharmaceutical-grade market as well.
The majority of sales are conducted either online, sold to customers in prohibition states or both. The ease and discretion of access for patients is a key selling point for hemp-derived CBD, and why existing products will not face significant competition from an expensive, highly regulated and narrowly prescribed product like Epidiolex in the immediate term.
 Bethany Gomez, Director of Research at Brightfield Group
Bethany Gomez, Director of Research at Brightfield Group
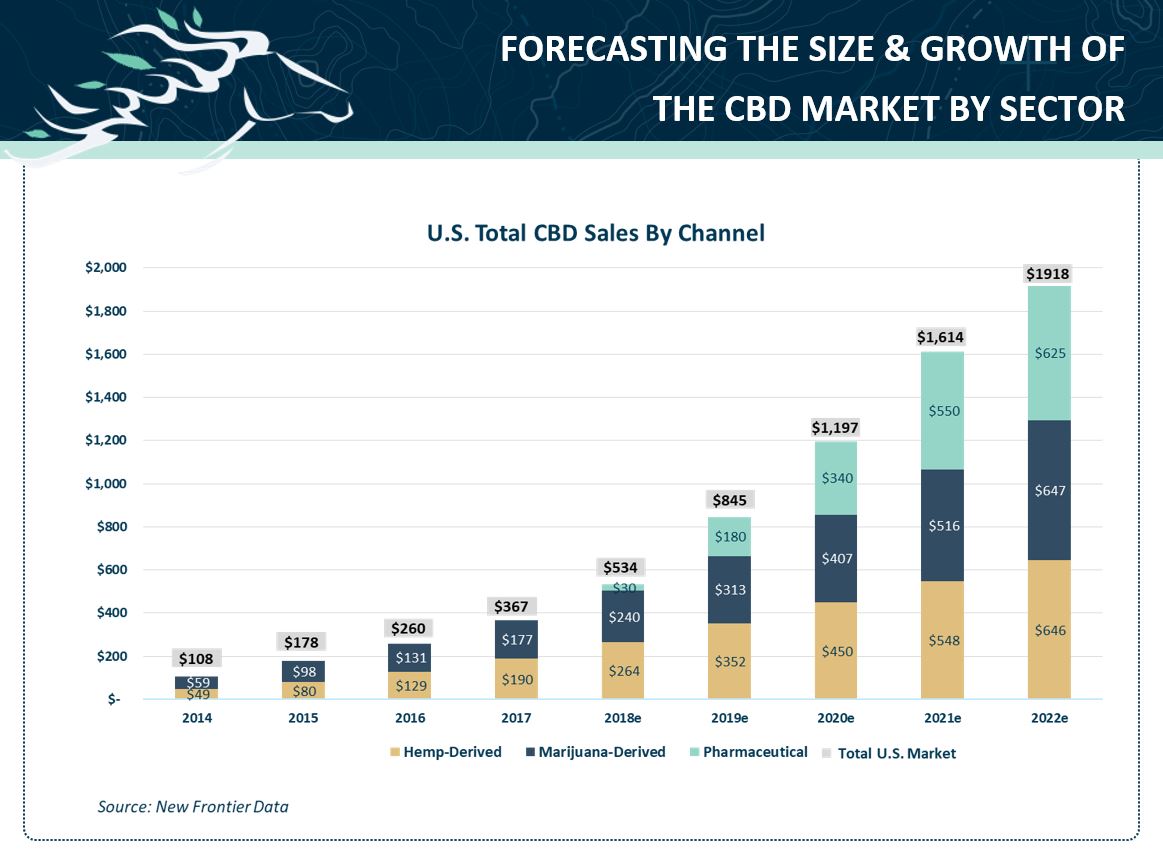
FDA approval of a CBD-based drug is likely to be a boon for the entire category, raising interest and acceptance of the cannabinoid as being safe and therapeutic. New Frontier Data predicts that the overall U.S. CBD market will increase more than 422% over the next five years to $1.9 billion:
A substantial portion of the projected growth will come from the pharmaceutical channel (Epidiolex, which New Frontier projects could reach $625 million in 2022), but even excluding Epidiolex, the data firm projects continued growth, with hemp-derived products increasing 240% and marijuana-derived products growing 265%. Several analysts are even more optimistic than New Frontier on the prospects for the drug, with some suggesting peak sales could exceed $1 billion.
While there have been some concerns in the industry that GW Pharma might take action to block the sale of CBD by other providers upon approval of Epidiolex by the FDA, we believe this is not the case based on many discussions we have had with the company over the years. Spokesperson Steven Schultz suggested that the goal of GW Pharma is to offer a pharmaceutical alternative to the existing market, one that would allow patients to obtain Epidiolex under physician care and through pharmacies, similar to other FDA-approved medicines. Sellers of CBD already face a risk of enforcement by the FDA if they make unsubstantiated health claims. Most recently, the FDA issued warning letters last year to several companies marketing CBD products for treatment or prevention of cancer. We expect that GW Pharma could pursue CBD product suppliers who make health claims with respect to epilepsy.
Attorney Bob Hoban of Hoban Law Group sees limited impact to dispensaries selling cannabis-based products or to sellers of CBD from industrial hemp.
As the first marijuana-derived drug set to be approved by FDA, the cannabis industry should closely monitor the progress of FDA’s review of Epidiolex. Given that the state-legal marijuana industry exists in violation of federal law already, the federal approval of Epidiolex should have little immediate impact on these businesses.
 Bob Hoban, Managing Partner at Hoban Law Group
Bob Hoban, Managing Partner at Hoban Law Group
On the other hand, industrial hemp businesses should be acutely aware that FDA’s review of Epidiolex may have a more immediate impact on the distinction of hemp-derived nutritional supplements — as are widely sold in the U.S. today — and Epidiolex. We anticipate Epidiolex to be an additional method of access, versus displacing the current methods of access, but are working tirelessly to ensure appropriate distinctions are made between drug, marijuana and hemp products all of which contain cannabinoids.
Two leading suppliers of CBD from industrial hemp, Elixinol Global (ASX: EXL) (OTC: ELLXF) and CV Sciences (OTC: CVSI), are encouraged by recent developments. Elixinol produces its CBD from hemp grown in Colorado, while CV Sciences imports the CBD from Europe.
With the combination of the pending FDA approval of Epidiolex and the recent momentum of the Hemp Farming Act, we are seeing simultaneous confirmation of both the healing properties of CBD as well as the legislative push to allow its sale and distribution as a botanical ingredient throughout the United States.
 Gabriel Ettenson, Co-Founder of Elixinol Global
Gabriel Ettenson, Co-Founder of Elixinol Global
These two elements, missing from an already rapidly growing industry, will likely combine to add significant fuel to the industry and promote strong growth domestically and internationally.
CV Sciences, which is also pursuing the FDA regulatory path for a chewing gum with nicotine and synthetic CBD, CVS-007, targeting smokeless tobacco sensation, is also optimistic.
We believe the FDA’s advisory committee approval of Epidiolex validates the tremendous potential of cannabidiol to improve patient outcomes. We look forward to advancing our own CBD-based drug development program for treatment of nicotine addiction.
 Joseph Dowling, CFO at CV Sciences
Joseph Dowling, CFO at CV Sciences
We also believe this approval strengthens the position of hemp extracts as the fastest growing category in the dietary supplement market.
Finally, state-legal cannabis companies could benefit as well from the FDA’s expected approval of a cannabis-derived medicine. According to Charles Bachtell, CEO of multi-state cannabis operator Cresco Labs, “While Epidiolex is a CBD-focused medicine, progress in any area of medical cannabis research is progress for cannabis in general. Now it would not only be irresponsible, but clinically inaccurate, to disregard the concept of cannabis having medicinal value — once you have an FDA approved plant-derived medication, that discussion is finally over.”
Conclusions
Approval of Epidiolex could have profound implications. While cannabis or even CBD aren’t required to be rescheduled in order for for it to be approved and then marketed by GW Pharma, the approval by the FDA of a CBD product derived from cannabis plants could play a role in the scheduling status of CBD or cannabis in the future.
We believe that the landmark approval of a cannabis-derived drug will not only spur further research and development of FDA-approved cannabis products but also accelerate an already rapidly growing market for the health and wellness market for CBD products, which are derived from industrial hemp and cannabis plants and sold in dispensaries, mail-order or other channels. The recently introduced Hemp Farming Act of 2018, supported by sponsor Senate Majority Leader Mitch McConnell, is likely to clarify some of the legal issues that suppliers of CBD products from industrial hemp have faced since the 2014 Farm Act opened the door to domestic hemp as a source of CBD.
At the same time, we expect a significant shakeout in the supply base of these non-pharmaceutical products. In early 2016, the FDA took action against eight companies marketing CBD products as dietary supplements for making health claims. The FDA website includes 40 separate warning letters sent to companies since 2015, some of which accused providers of selling products with little or no CBD. Companies that succeed are likely to be the ones that have high quality control standards and marketing practices that steer clear of FDA scrutiny, while those pushing snake oil will likely struggle.
The bottom-line, then, is that approval of Epidiolex is likely to be a boon for patients suffering from epilepsy and to open the door to a pharmaceutical distribution pathway for cannabis-derived drugs, but it is also likely to drive significant interest in the broader CBD category. In the same way that the over-the-counter fish-oil market has thrived more than a decade after the introduction of prescription-strength products, the CBD market is likely to see significant expansion in the coming years.